ORCHID
ORCHID (INA-107) : INA-RESPOND Observational Research on Infectious Disease Outbreaks & Difficult Cases of Unidentified Etiology in Indonesia; COVID-19 Pandemic
ClinicalTrials.gov: NCT04339179
Collaborator: NIAID, NIH
Status: Completed
https://clinicaltrials.gov/study/NCT04339179
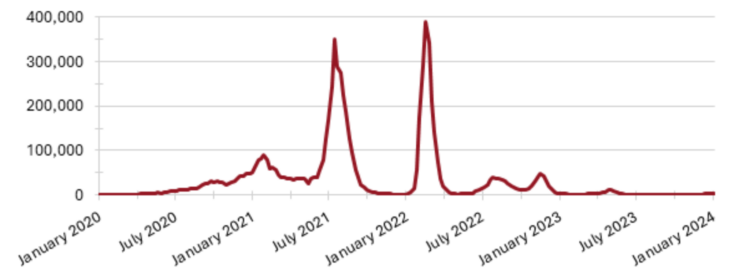
The ORCHID observational study was originally designed to prospectively investigate infections of unknown etiology during outbreaks and in challenging hospitalization cases, with a focus on emerging and re-emerging diseases. The study was quickly repurposed during the COVID-19 pandemic to focus on patients hospitalized with suspected SARS-CoV-2 infection. From November 2020 to July 2022, 185 adults were enrolled and followed for at least 14 days. All participants provided blood, respiratory swabs, urine, faeces, and saliva for on-site and centralized testing, including SARS-CoV-2 rapid diagnostic test, RT-PCR, and serology; acid-fast bacilli smear; blood culture; typhoid rapid diagnostic test; influenza rapid diagnostic test; and dengue rapid diagnostic test, RT-PCR, and serology. Among the participants, 152 had SARS-CoV-2, including 2 with concurrent dengue infection. Other detected pathogens included dengue (4), M. tuberculosis (1), influenza A and B (3, 1), respiratory syncytial virus (1), and Haemophilus influenzae type B and non-type B (1, 6). Only 61 (33.3%) participants had received at least one dose of a COVID-19 vaccine before enrollment, mostly (72.1%) from Sinovac, and 5 (2.7%) participants on the study died.
Associated Biorepository
| Specimen Type | Baseline | Follow up (7-14 days) | ||
| COVID-19 Positive | COVID-19 Negative | COVID-19 Positive | COVID-19 Negative | |
| Plasma EDTA | 148 | 32 | N/A | N/A |
| Buffy Coat | 121 | 30 | N/A | N/A |
| Serum | 149 | 32 | 137 | 11 |
| Swab (NP) in VTM | 149 | 32 | 139 | 10 |
| Swab (OP) in VTM | 149 | 32 | 139 | 10 |
| Urine | 137 | 30 | 103 | 7 |
| Stool | 95 | 18 | 44 | 3 |
| Saliva | 116 | 25 | 86 | 7 |
| Specimen Type | Visit | Variant* | ||||||||||
| Delta | Omicron | Other | ||||||||||
| AY.23 | AY.24 | B.1.617.2 | BA.1 | BA.1.1 | BA.1.13.1 | B.1 | B.1.1.398 | B.1.459 | B.1.466 | B.1.466.2 | ||
| Plasma EDTA | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 7 | 5 | 12 | 34 |
| Buffy Coat | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 0 | 0 | 11 | 28 |
| Serum | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 8 | 5 | 12 | 34 |
| Day 7-14 | 12 | 3 | 3 | 1 | 1 | 4 | 1 | 8 | 5 | 12 | 34 | |
| Urine | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 8 | 4 | 10 | 30 |
| Day 7-14 | 12 | 3 | 3 | 1 | 1 | 2 | 1 | 6 | 3 | 10 | 28 | |
| Faeces | Enroll. | 9 | 4 | 1 | 1 | 1 | 2 | 1 | 5 | 4 | 8 | 18 |
| Day 7-14 | 3 | 1 | 2 | 0 | 0 | 1 | 1 | 3 | 3 | 7 | 8 | |
| Saliva | Enroll. | 12 | 4 | 3 | 1 | 2 | 3 | 1 | 7 | 2 | 10 | 23 |
| Day 7-14 | 12 | 2 | 3 | 0 | 0 | 1 | 1 | 4 | 1 | 9 | 26 | |
| Swab (NP) in VTM | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 8 | 5 | 12 | 34 |
| Day 7-14 | 13 | 3 | 3 | 1 | 1 | 4 | 1 | 8 | 5 | 12 | 34 | |
| Swab (OP) in VTM | Enroll. | 13 | 5 | 3 | 1 | 2 | 5 | 1 | 8 | 5 | 12 | 34 |
| Day 7-14 | 13 | 3 | 3 | 1 | 1 | 4 | 1 | 8 | 5 | 12 | 34 | |
| *Sequencing data are available for 89 participants (submitted to GISAID). Accession IDs are EPI_ISL_19750669 to EPI_ISL_19750753, and EPI_ISL_19761433 to EPI_ISL_19761436. | ||||||||||||
