ITAC
ITAC/INSIGHT-013 : An International Multicentre, Adaptive, Randomized Double-Blind, Placebo-Controlled Trial of the Safety, Tolerability and Efficacy of Anti Coronavirus Hyperimmune Intravenous lmmunoglobulin for the Treatment of Adult Hospitalized Patients at Onset of Clinical Progression of COVID-19 / Inpatient Treatment with Anti-Coronavirus lmmunoglobulin
ClinicalTrials.gov: NCT04546581
Collaborator: NIAID, NIH
Status: Completed
https://clinicaltrials.gov/study/NCT04546581
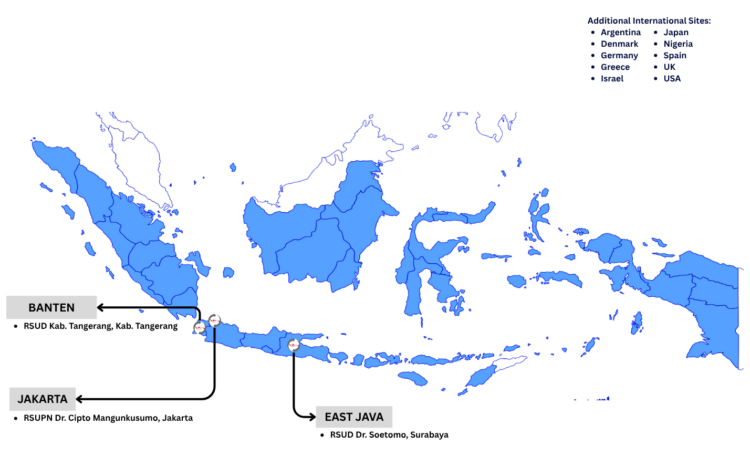
The ITAC study was an international, double-blind, placebo-controlled, phase 3 trial evaluating the safety and clinical efficacy of a single infusion of anti-SARS-CoV-2 hyperimmune intravenous immunoglobulin in individuals hospitalized with COVID-19 without end-organ failure. The trial enrolled 593 adults between October 2020 and March 2021. Sites in Indonesia contributed 33 participants between January and February 2021. Participants were followed for 28 days after randomization and provided a nasal swab at baseline and blood at timepoints throughout the study. Specimens were used to determine SARS-CoV-2 viral RNA levels, SARS-CoV-2 nucleocapsid antigen levels, anti-spike receptor binding domain neutralizing antibody levels, and anti-nucleocapsid binding antibody levels. The primary endpoint was an ordinal outcome based on participant clinical status on day 7, and a modified intention to treat analysis found that participants randomized to the intervention group did not have significantly greater odds of a more favorable outcome at day 7 compared to the placebo group.
Associated Publications:
- Hyperimmune immunoglobulin for hospitalised patients with COVID-19 (ITAC): a double-blind, placebo-controlled, phase 3, randomised trial. Lancet. 2022 Feb 5;399(10324):530-540. doi: 10.1016/S0140-6736(22)00101-5. Epub 2022 Jan 28. PMID: 35093205.
Associated Biorepository
| Specimen Type | Baseline | Day 1 | Day 2 | Day 3 | Day 7 | Day 28 | Day 90 |
| Plasma | 28 | 33 | 32 | 32 | 33 | 26 | 31 |
| Serum | 28 | 33 | 32 | 32 | 33 | 26 | 31 |
| Plasma | 33 | N/A | N/A | N/A | N/A | N/A | N/A |
