SchisCCA
SchisCCA (INA-105) : Validation of the Schistosomiasis Point-of-Care Circulating Cathodic Antigen {POC-CCA} Rapid Urine Test for Qualitative Detection of Schistosoma japonicum
ClinicalTrials.gov: NCT03870204
Collaborator: NIAID, NIH
Status: Completed
https://clinicaltrials.gov/study/NCT03870204
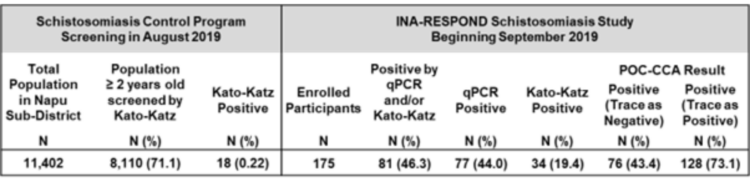
The SchisCCA longitudinal cohort study was conducted in Napu Valley, Central Sulawesi, between September 2019 and February 2020 in coordination with the 2019 national schistosomiasis stool survey and mass drug administration campaign. Data from the 2018 and 2019 surveys informed the targeted enrollment of 185 participants (124 adults, 61 children) who had a history of infection with S. japonicum. Demographic information, clinical data, urine, and three consecutive stool specimens were collected from each participant after enrollment. Urine was used in a point of care schistosomiasis diagnostic test (POC-CCA) to evaluate sensitivity and specificity against gold standard Kato-Katz stool microscopy and centralized stool qPCR testing. Participants who were positive for schistosomiasis received standard treatment at a follow-up visit and provided an additional set of three stool specimens after 8 weeks to observe instances of reinfection. Microscopy revealed an infection prevalence of 19.4%, which was much lower than qPCR (44.0%) and POC-CCA (43.4% or 73.1%, depending on the strictness of test interpretation). The POC-CCA test had the lowest accuracy and consistently overestimated infection prevalence.
Associated Biorepository
| Testing type | Baseline | Follow up | ||
| (Visit 3/Post-Treatment) | ||||
| Positive | Negative | Positive | Negative | |
| Stool PCR | 81 | 102 | 15 | 28 |
| Stool Kato Katz | 29 | 156 | 26 | 18 |
| Specimen type | Baseline | Follow up |
| (Visit 3/Post-Treatment) | ||
| Stool DNA extraction | 185 | 44 |
