
ORAL ANTIVIRUS FOR COVID-19
The coronavirus disease 2019 (COVID-19) pandemic continues to grow. Protective vaccines have been developed within unprecedented timelines, but current supplies are too low to cover worldwide demand in the coming months. Moreover, a significant number of people are either unable, due to pre-existing medical conditions, or unwilling to be vaccinated, and global access challenges remain. Unlike vaccines that can prevent infection, antivirals act as a second line of defense. Furthermore, SARS-CoV-2 is likely to become endemic, leading to the emergence of vaccine-resistant variants and reinforcing the need to develop antiviral therapeutic agents. Researchers worldwide are urgently looking for interventions to prevent new infections, prevent disease progression, and lessen disease severity for those already infected. SARS-CoV-2 specific therapeutics are urgently needed to prevent more severe disease, hospitalization, and death. Treatment may also reduce the period of infectivity.
Development of Antiviral for Covid-19
Developing the new antivirals is an expensive and difficult endeavor, especially for acute respiratory diseases, for which the window for treatment is short. Historically antiviral drug development has focused on a “one bug, one drug” approach, targeting proteins common to specific groups of viruses. Researchers started screening molecular collections, such as the California Institute for Biomedical Research’s ReFRAME, to test if any FDA-approved drugs and investigational compounds were effective against SARS-CoV-2. Laura Riva, a computational biologist formerly at the Sanford Burnham Prebys Medical Discovery Institute in California, conducted one such screen along with her colleagues and identified more than a dozen compounds, including remdesivir, that blocked SARS-CoV-2 replication in animal and human cells (1). While such antivirals can be extremely effective, viruses produce very few proteins of their own, giving drug makers limited options. There’s also the risk of the drugs damaging cells. Some viral proteins can be unique in that they don’t overlap with the ones produced by the host, making them ideal targets for antiviral drugs. But if the target proteins do overlap or perform the same functions as the human host cells, there is potential for collateral damage, resulting in side effects. Once drugmakers have identified a target, the compound has to go through a lengthy testing phase. The first step involves demonstrating that the compound works on infected cells in Petri dishes, then assessing if it is safe and effective in laboratory animals, and finally in human clinical trials. Therefore, normally making antiviral therapies for new viruses can take at least a decade (2). However, the urgency presented by COVID-19 meant finding new ways to use old drugs.
While research on new therapeutic agents for COVID-19 is key, there is also great interest in evaluating the potential of already existing medicines against COVID-19. The urgency amid the pandemic has caused interest in repurposing other drugs targeted for other diseases. The known safety profiles, shortened development timelines, and well-established markets for most of the already existing compounds proposed for COVID-19 are particularly advantageous compared to new drug discovery in a pandemic situation. However, repurposing approved drugs in the search for small molecule antiviral agents that target SARS-CoV-2 has thus far been minimally effective (3). So far, Gilead’s remdesivir, originally developed for Ebola infections, is the only such repurposed antiviral drug that has received approval from the U.S. Food and Drug Administration to treat hospitalized patients with COVID-19 (4). When used in a hospital setting, its effect is modest. In a phase 3 trial, researchers found that it shortened recovery time by a median of 5 days (5). However, its wider use is limited by intravenous delivery due to the limited oral bioavailability. The therapeutic benefits of remdesivir are also under ongoing debate as WHO has issued a conditional recommendation against the use of remdesivir in hospitalized patients, regardless of disease severity, as there is currently no evidence that remdesivir improves survival and other outcomes in these patients based on the results of multinational SOLIDARITY trial (6).
Other than Remdesivir, three anti-SARS-CoV-2 monoclonal antibodies (mAbs) that target the spike protein has been shown to have a clinical benefit SARS-CoV-2 infection. During the infection of host cells by SARS-CoV-2, the spike (S) glycoprotein of SARS-CoV-2 plays the most crucial role in viral entry and cell fusion. The spike protein is further divided into S1 and S2, which mediate host cell attachment and invasion. Through its receptor-binding domain (RBD), S1 attaches to angiotensin-converting enzyme 2 (ACE2) on the host cell; this initiates a conformational change in S2 that results in virus-host cell membrane fusion and viral entry. The mAbs can specifically bind to SARS-CoV-2 RBD, block the interaction between SARS-CoV-2 RBD and the human ACE2 receptor, and thus block viral attachment and entry into human cells, leading to efficient neutralization of the virus (7). Studies have shown that MAbs are effective in preventing SARS-CoV-2 infection in household contacts of infected patients and during SARS-CoV-2 outbreaks in skilled nursing and assisted living facilities, which can cut the risk of hospitalization and death by up to 85 percent (8). The three Mab products (Bamlanivinab plus etesevimab, Casirivimab plus imdevimab, and Sotrovimab) have received Emergency Use Authorizations (EUAs) from the Food and Drug Administration (FDA) for the treatment of mild to moderate COVID-19 in non-hospitalized patients with laboratory-confirmed SARS-CoV-2 infection who are at high risk for progressing to severe disease and/or hospitalization (9). Despite the benefits, mAbs are costly, must be administered through an IV in a medical setting, and, in poor nations, scarce or absent. Yet without widespread vaccination, those populations remain vulnerable to COVID-19 and need affordable medicines.
As an acute viral infection triggers COVID-19, the antiviral therapeutics will be most effective if given within the early stages of the infection when viral load is at its maximum, during rapid replication of SARS-CoV-2 in nasopharyngeal and respiratory epithelium (10). Thus, an orally available direct-acting antiviral would be essential for such treatment in an outpatient setting. A new generation of orally available broad-spectrum antivirals is emerging that should allow initiation of treatment early after infection and prevent further systemic dissemination of the virus and development of systemic inflammation. A pill could make treating patients earlier in their infection much easier — and more effective than intravenous treatment, which requires certain medical resources for infusion and patient monitoring at the hospital. With the medical system under intense pressure, oral antiviral drugs that can reduce the viral burden from the initial stages of infection and are easy to use are required. This therapeutic agent is expected to contribute to patients’ early treatment and help keep hospitals from overflowing and thus relieve the pressure on the medical system (11).
How the antiviral works
The development of effective intervention strategies relies on the knowledge of molecular and cellular mechanisms of coronavirus infections, which highlights the significance of studying virus-host interactions at the molecular level to identify targets for antiviral intervention and to elucidate critical viral and host determinants that are decisive for the development of severe disease. During the intracellular life cycle (Fig. 1), coronaviruses express and replicate their genomic RNA to produce full-length copies that are incorporated into newly produced viral particles. Upon the entry, the uncoating by nucleocapsid degradation allows the release into the cytoplasm of the viral RNA, ready for translation. The 5′- and 3′-UTRs flank the coding region with the two-thirds of the genome from the 5′-end comprising two overlapping open reading frames (ORFs), ORF1a and ORF1b, that encode for polyproteins pp1a (4382 amino acids) and pp1ab (7073 amino acids), respectively. The autoproteolytically processing by 3CL or Main (Mpro) and PLpro affords 16 nonstructural proteins (nsp1–16), which form the replicase/transcriptase complex (RTC). The RTC includes different enzymes and cofactors involved in post-translational polyprotein processing, RNA synthesis, maturation, and virions assembly and egress, which therefore can constitute ideal viral targets for drug discovery, being essential for the virus life cycle and devoid of a close host homologue. Genomic ss-(+)-RNA transcription proceeds through (−)-strand intermediates that serve as templates for the production of both genomic and subgenomic RNAs, which are capped and polyadenylated as the full genomic RNA. The subgenomic RNAs are then translated into the four structural and some accessory proteins (12). On the basis of the prominent roles in intracellular steps of viral life cycle, the amount of biochemical/structural data and the knowledge acquired on inhibitors of homologues proteins in other CoVs and other RNA viruses, the 3CLpro and the nsp12 RdRp are at moment the most relevant viral targets to identify specific anti-CoVs agents (13).
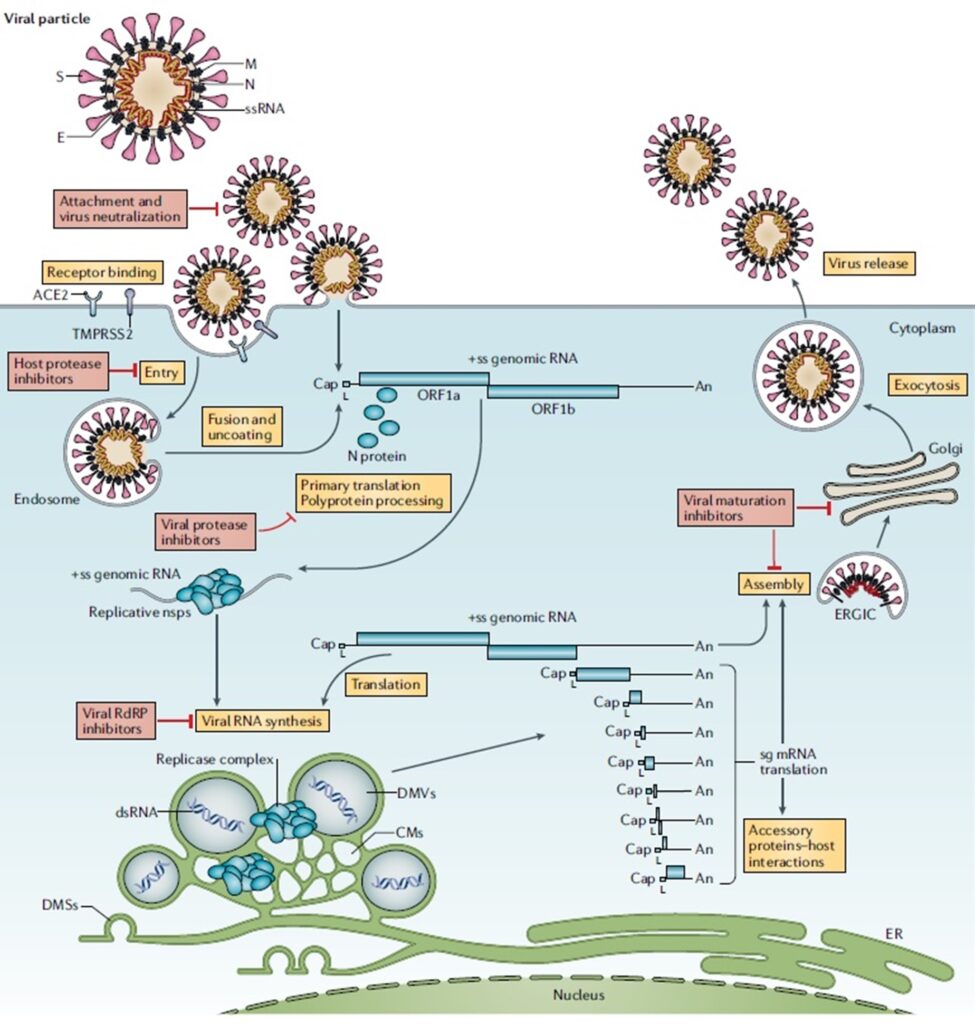
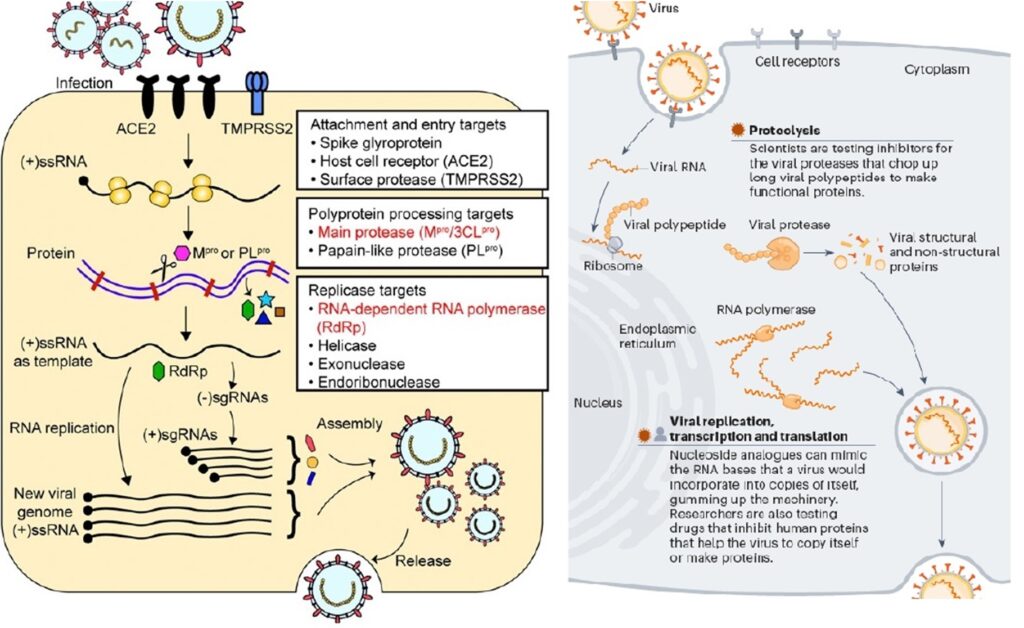
Previous research efforts to develop antiviral agents against the members of the coronavirus family suggested the ACE2 entry receptor, the RNA-dependent RNA polymerase (RdRp), and the main protease (Mpro) as suitable drug targets (Fig. 2). As there is a high chance that coronaviruses will undergo mutations to become a new infectious virus, identification of promising targets for antiviral therapies against SARS-CoV-2 should exploit the structural similarities among different coronaviruses and focus on those proteins that are highly conserved across multiple coronaviruses. Among the several potential targets of coronaviruses, replication-related enzymes, such as RdRp and protease, are highly conserved. Drugs that inhibit conserved proteases, such as Mpro and papain-like protease (PLpro), can prevent replication and proliferation of the virus by interfering with the posttranslational processing of essential viral polypeptides and can also reduce the risk of mutation-mediated drug resistance. Replication of SARS-CoV-2 depends on RNA-dependent RNA polymerase (RdRp), and thus RdRp is also a promising drug target for the treatment of coronaviruses (14). Remdesivir has shown antiviral activity against SARS-CoV-2 in vivo in rhesus monkeys through the targeting of RdRp (15). Computational drug repurposing is an effective approach to find new indications for the drugs already approved for other functions. This virtual drug screening strategy, comprising the pre-docking filtering, docking simulation, and post-docking filtering processes, was applied to identify drug candidates targeting two key enzymes of SARS-CoV-2, Mpro and RdRp, using their crystal or cryoelectron microscopy (cryo-EM) structures (14).
Investigational oral antiviral candidates for Covid-19
1. Molnupiravir
Molnupiravir (MK-4482/EIDD-2801) is an investigational, originally designed to fight the flu, orally administered form of a potent ribonucleoside analog that inhibits the replication of SARS-CoV-2. Molnupiravir has been shown to be effective in several preclinical models of SARS-CoV-2, including for prophylaxis, treatment, and prevention of transmission. Like remdesivir, molnupiravir targets the RdRp, which mediates replication and transcription of the coronavirus genome, leading to increased frequency of G-to-A and C-to-U transition mutations (Fig. 3). This will disrupt the fidelity of SARS-CoV-2 genome replication and prevents viral propagation by fostering error accumulation in a process referred to as ‘error catastrophe’, which forces the SARS-CoV-2coronavirus to mutate itself to death (lethal mutagenesis). Molnupiravir was also shown to inhibit propagation of the SARS-CoV, MERS-CoV, and SARS-CoV-2 viruses, re-enforcing its pan-coronaviral inhibitory profile. Treatment with molnupiravir failed to induce viral-resistance mutations, which suggests a high genetic barrier to immune evasion (16).
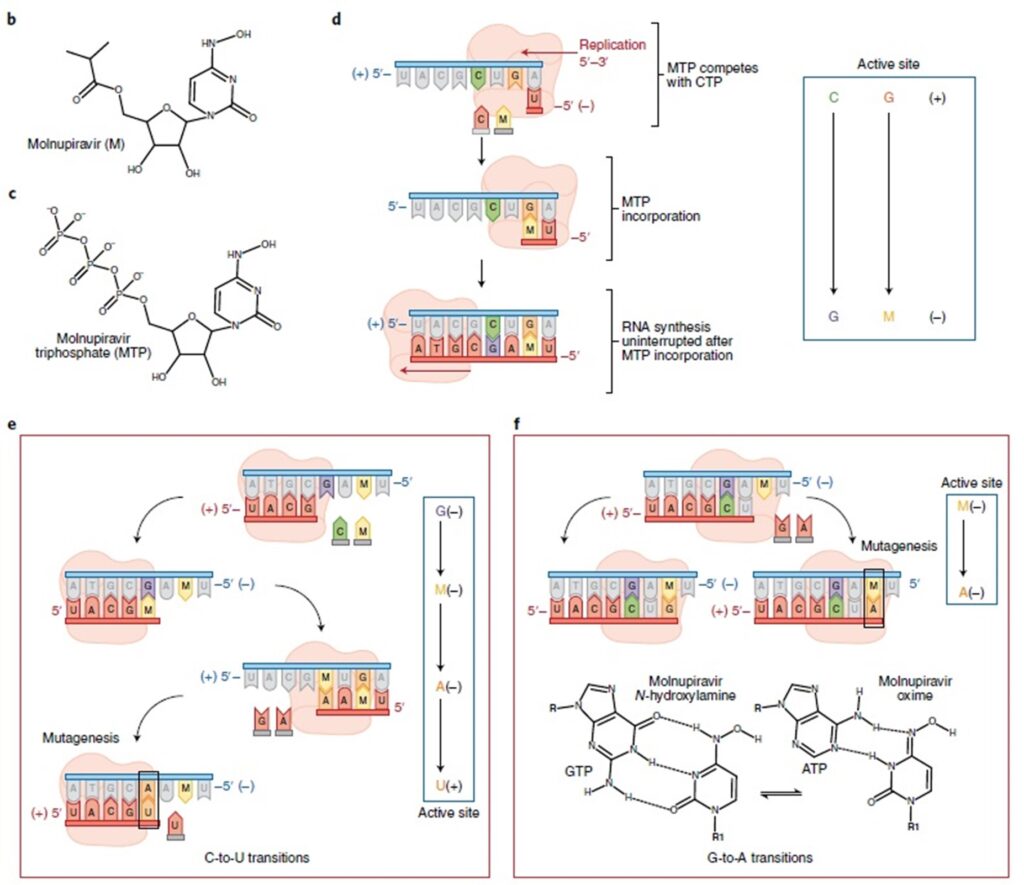
The accumulation of mutations pushes viral replication over the ‘error threshold’ that demarcates the replication fidelity required for viability. This mechanism distinguishes Molnupiravir from remdesivir, which impedes the progression of viral RdRp, and provides insights into alternative mechanisms of RdRp inhibition. Finally, Molnupiravir possesses excellent pharmacokinetic properties, which include oral administration. An orally bioavailable antiviral will have far-reaching benefits in tackling the spread of COVID-19 in hard-to-reach communities worldwide. As with all therapeutic agents, off-target effects are a concern. In its triphosphate form, Molnupirivar is a substrate for the mitochondrial RNA polymerase, which can also incorporate MTP as a U or C analog. Reassuringly, the study noted that mitochondrial function over 14 days was not significantly inhibited, and another study did not observe mutagenesis of host mRNA. However, it has been suggested that exposure to Molnupiravir can be mutagenic to host DNA during host DNA replication. Therefore, the potential off-target effects will require further investigation (11,16).
In their press release last October, Merck announced that Molnupiravir significantly reduced the risk of hospitalization or death among people with COVID-19 based on an interim analysis of Phase 3 MOVe-OUT trial in at risk, enrolling non-hospitalized adult patients with mild-to-moderate COVID-19. At the interim analysis, molnupiravir reduced the risk of hospitalization or death by approximately 50%; 7.3% of patients who received molnupiravir were either hospitalized or died through Day 29 following randomization (28/385), compared with 14.1% of placebo-treated patients (53/377); p=0.0012. In addition, through Day 29, no deaths were reported in patients who received molnupiravir compared to 8 deaths in patients who received placebo. At the recommendation of an independent Data Monitoring Committee and consultation with the U.S. FDA, recruitment into the study is being stopped early due to these positive results (17). Although interim findings aren’t yet peer-reviewed, the companies jointly applied for an emergency use FDA authorization of the pill on October 11; while the U.K. has authorized Molnupirvair’s use on November 4. The drug needs to be given needs to be given within the first five days of symptoms onset, at about $700 per tx course (twice daily for five days).
2. PAXLOVIDTM (PF-07321332; ritonavir)
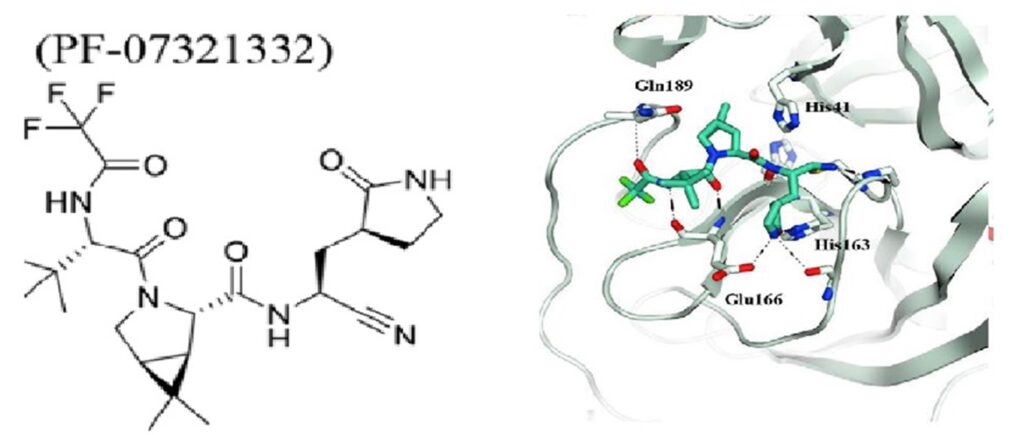
PAXLOVID™ is an investigational SARS-CoV-2 protease inhibitor antiviral therapy, specifically designed to be administered orally to be prescribed at the first sign of infection, potentially helping patients avoid severe illness, hospitalization, and death. Pfizer developed PF-07321332 in early 2000s as a potential treatment for SARS caused by the coronavirus SARS-CoV. At the start of the Covid-19 pandemic, they retooled it to work against SARS-CoV-2, which has similar biology with SARS-CoV. In addition, they modified the drug, originally designed to be given intravenously, as a pill. Pfizer’s PF-07321332 is designed to block the activity of the SARS-CoV-2-3CL protease (Mpro) (Fig.4), thus inhibit proteolysis, which occurs before viral RNA replication. Co-administration with a low dose of HIV antiviral ritonavir helps slow the metabolism, or breakdown, of PF-07321332 to remain active in the body for longer periods at higher concentrations to help combat the virus. In pre-clinical studies, PF 07321332 did not demonstrate evidence of mutagenic DNA interactions (18).
PAXLOVID™, as the drug is now known, went into clinical trials in March 2021, followed by a larger Phase 3 trial in July. On November 5, based on an interim analysis of the Phase 2/3 EPIC-HR (Evaluation of Protease Inhibition for COVID-19 in High-Risk Patients) a randomized, double-blind study of non-hospitalized adult patients with COVID-19, the administration of this drug showed an 89% reduction in risk of COVID-19-related hospitalization or death from any cause compared to placebo in patients treated within three days of symptom onset (primary endpoint); 0.8% of patients who received PAXLOVID™ were hospitalized through Day 28 following randomization (3/389 hospitalized with no deaths), compared to 7.0% of patients who received placebo and were hospitalized or died (27/385 hospitalized with seven subsequent deaths). The statistical significance of these results was high (p<0.0001). Similar reductions in COVID-19-related hospitalization or death were observed in patients treated within five days of symptom onset; 1.0% of patients who received PAXLOVID™ were hospitalized through Day 28 following randomization (6/607 hospitalized, with no deaths), compared to 6.7% of patients who received a placebo (41/612 hospitalized with ten subsequent deaths), with high statistical significance (p<0.0001). In addition, in the overall study population through Day 28, no deaths were reported in patients who received PAXLOVID™ as compared to 10 (1.6%) deaths in patients who received placebo (19). At the recommendation of an independent Data Monitoring Committee and in consultation with the U.S. FDA, Pfizer will cease further enrollment into the study due to the overwhelming efficacy demonstrated in these results and plans to submit the data as part of its ongoing rolling submission to the U.S. FDA for EUA as soon as possible. Similar with Molnupiravir, PAXLOVID™ needs to be given within the first five days of symptoms onset, at about $700 per treatment course (twice daily for five days).
3. AT-527
An oral direct-acting antiviral, AT-527, is being co-developed by Atea Pharmaceuticals in partnership with Roche. AT-527, a double prodrug of a guanosine nucleotide analog, was previously shown to be highly efficacious and well-tolerated in hepatitis C virus (HCV)-infected subjects. Its unique mechanism of action, with dual targets including chain termination of RdRp and nidovirus RdRp associated nucleotidyltransferase (NiRAN), which serves to prime the RdRp for RNA synthesis inhibition, and thus has the potential to create a high barrier to resistance with broad antiviral coverage to different variants of SARS-CoV-2 (Fig. 5). Atea has completed a comprehensive nonclinical program to characterize the potency and safety profile of AT-527. In vitro analysis showed potent antiviral activity against flaviviruses and coronaviruses, including SARS-CoV-2 (EC90=0.5 µM). Results from these nonclinical studies demonstrate that AT-527 is non-mutagenic and has no effects on fertility and reproduction (20).
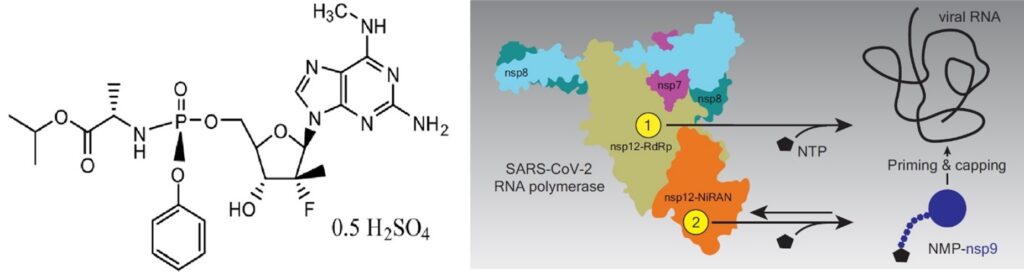
Atea Pharmaceuticals in its October press release provided update and topline results for Phase 2 MOONSONG Trial Evaluating AT-527 in the outpatient setting. The randomised, multicentre, double-blind, placebo-controlled Phase II trial assessed the safety, antiviral activity and pharmacokinetics of twice-daily (BID) doses of 550 mg and 1,100 mg AT-527 in adult subjects with mild or moderate Covid-19 against placebo. Findings showed that AT-527 failed to meet the primary goal versus placebo in the overall trial population, which had two-thirds of subjects with mild symptoms at reduced risk. A decline of viral load of nearly 0.5log₁₀ at day seven was reported in increased risk subjects with underlying health conditions when administered with BID 550 mg and 1,100 mg AT-527 against placebo. Nearly 20% in the placebo arm and AT-527 550mg BID arm reported adverse event (AE) versus 27% in the AT-527 1100 mg BID arm. Furthermore, the most common AEs observed in the trial were gastrointestinal-related (21). “Based on the totality of the results for AT-527 to-date, the current level of understanding of the virus and the evolving COVID-19 environment, we are assessing the Phase 3 MORNINGSKY trial for modifications to ensure the best possible outcome for the program,” said Janet Hammond, MD, PhD, Chief Development Officer of Atea Pharmaceuticals. “We, along with our partner Roche, are continuing to advance multiple studies in parallel to provide further clinical evidence as well as outcome data to support AT-527 as an oral, potent, direct-acting antiviral treatment for COVID-19.” Possible changes to the international Phase III MORNINGSKY trial, including its primary goal and subject population, will be analyzed by Atea and Roche. Results from this trial are anticipated in the second half of next year (2022).
4. Favipiravir
Favipiravir (6-fluoro-3-hydroxy-2-pyrazinecarboxamide) also known as T-705, avigan, or favilavir, is an antiviral agent that was first developed by the Japanese Toyama Chemical Co., a subsidiary of Fuji Film. It was originally designed to beat against the flu, as reported initially by Furuta in 2002. In 2014, it was approved in Japan as a backup choice for resistant influenza infection. Favipiravir is a pyrazinecarboxamide derivative that can inhibit the RdRp of influenza virus and has also been shown to have antiviral activities beyond the flu. In animals, favipiravir has shown activity against viruses such as Influenza, West Nile, Yellow fever, Foot and Mouth Disease, and Rift Valley as well as other Flaviviruses, Arenaviruses, Bunyaviruses, Alphaviruses (e.g., Chikungunya [CHIKV]; Sindbis [SINV]; Western equine encephalitis [WEEV]; and Semliki Forest [SFV] viruses), and Enteroviruses. Favipiravir has been shown to have an effect in vitro and in vivo against Zaire Ebola, Rabies (RABV), and Zika viruses. Favipiravir was described to reduce the morbidity and mortality associated with RABV infection in mice. In a Syrian hamster model that mirrors the human disease, favipiravir reduced encephalitis, hemorrhagic fever, respiratory difficulties, and mortality rate caused by Nipah virus infection (a Bat virus) (22).
Favipiravir is structurally similar to ribavirin (antiviral drug used to treat Respiratory Syncytial Virus [RSV] infection, Hepatitis C, and some viral hemorrhagic fevers). Favipiravir and ribavirin share a carboxamide (C-[O]-NH2) moiety. However, favipiravir is a more specific version of ribavirin. Both drugs target the viral RNA polymerase; ribavirin primarily targets the Inosine-5′-monophosphate dehydrogenase (IMPDH), while favipiravir interacts with RNA polymerase. Mechanistically, favipiravir is a prodrug that does not inhibit influenza RNA polymerase activity until it is phosphoribosylated in cells forming favipiravir-ribofuranosyl-50-triphosphate (favipiravir-RTP). Favipiravir-RTP then binds to the active site of RdRp to stop RNA replication (23). It has been suggested that the human hypoxanthine guanine phosphoribosyl-transferase (HGPRT) plays a role in the favipiravir activation. The active form of favipiravir is recognized by the catalytic domain of the viral RNA-dependent RNA polymerase and blocks its enzymatic activity. This results in inhibition of the RNA-dependent RNA polymerase and effectively ending the infectious cycle of SARS-CoV-2 (Fig. 6). Note that favipiravir is not toxic to mammalian cells and does not inhibit RNA or DNA synthesis within these cells (22,24).
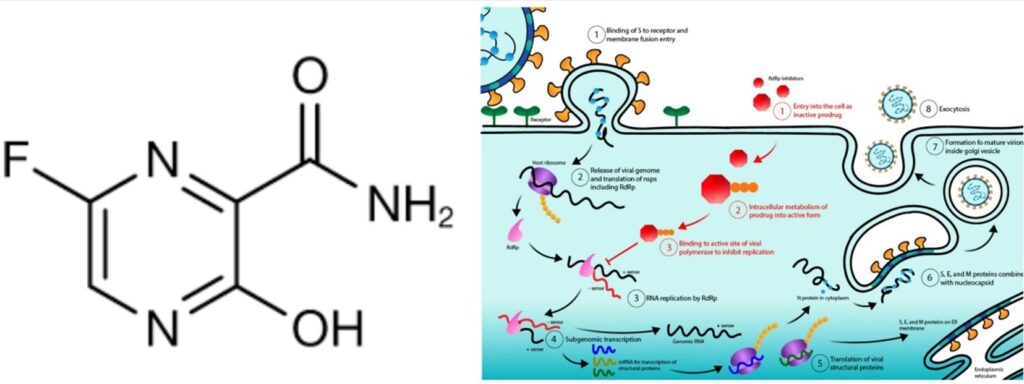
Previous favipiravir trials, albeit small, had suggested that in mild to moderate hospitalized COVID-19 patients, the drug could clear SARS-CoV-2 in their noses and throats, leading to a number of countries, including Japan, Kenya, Russia, Saudi Arabia, and Thailand, to approve favipiravir for Covid-19. But a February review of favipiravir trials suggested that it has only negligible impact on mortality in patients with serious symptoms. Nine studies encompassing 827 patients were included the meta-analysis (PROSPERO (CRD42020180032)) to determine the efficacy and safety of Favipiravir against COVID-19. The results revealed a significant clinical improvement in the Favipiravir group versus the control group during seven days after hospitalization (RR = 1.24, 95% CI: 1.09–1.41; P = 0.001). Viral clearance was more in 14 days after hospitalization in Favipiravir group than control group, but this finding was marginally not significant (RR = 1.11, 95% CI: 0.98–1.25; P = 0.094). Requiring supplemental oxygen therapy in the Favipiravir group was 7% less than the control group, (RR = 0.93, 95% CI: 0.67–1.28; P = 0.664). Transferred to ICU and adverse events were not statistically different between the two groups. The mortality rate in the Favipiravir group was approximately 30% less than the control group, but this finding was not statistically significant (25). Favipiravir possibly exerted no significant beneficial effect in terms of mortality in the general group of patients with mild to moderate COVID-19. Other trials are still underway to see if it can be useful for early treatment for people recently diagnosed with Covid-19, such as PRINCIPLE trial lead by University of Oxford UK (https://www.principletrial.org/news/favipiravir-to-be-investigated-as-a-possible-covid-19-treatment-for-at-home-recovery-in-the-principle-trial)
5. S-217622
S-217622, a therapeutic drug for COVID-19, is a 3CL protease inhibitor created through joint research between Hokkaido University and Shionogi & Co., Ltd. (Head Office: Osaka, Japan; President and CEO: Isao Teshirogi, Ph.D.; hereafter “Shionogi”). The new coronavirus (SARS-CoV-2) has an enzyme called 3CL protease, which is essential for the replication of the virus. S-217622 suppresses the replication of SARS-CoV-2 by selectively inhibiting 3CL protease. During at the International Society for Influenza and Other Respiratory Virus Diseases (ISIRV)-World Health Organization (WHO) Virtual Conference., the results of non-clinical drug efficacy and pharmacokinetic studies, and a summary of the results from the Japanese Phase 1 clinical trial1 which started in July 2021, were presented. The information presented is outlined below:
- Non-clinical studies using SARS-CoV-2 infected animals:
- S-217622 showed in vitro antiviral activity against a broad range of strains, including the δ strain.
- A dose-dependent viral reduction effect of S-217622 was observed in multiple animal studies.
- S-217622 showed a good drug metabolism and pharmacokinetics profile supporting oral dosing.
- The Japanese Phase 1 clinical trial (a single ascending dose study), began in July 2021:
- Single oral administration of S-217622 to healthy Japanese subjects was safe and well-tolerated.
- The once-daily oral dosing of S-217622 was predicted to exceed the target concentration required for the viral reduction effect from the non-clinical studies.
Based on these results, S-217622 has the potential to reduce SARS-CoV-2 viral load with once-daily oral administration. Phase 2/3 clinical trial of S-217622 is currently underway in mild or asymptomatic COVID-19 patients. The Phase 2/3 clinical trial will evaluate the efficacy and safety of oral administration of this drug once daily for five days in patients with mild COVID-19 or asymptomatic SARS-CoV-2 infection compared to placebo (26).
6. EDP-235
In August 2021, Enanta Pharmaceuticals, Inc. a clinical stage biotechnology company, announced that it had nominated EDP-235, its lead oral protease inhibitor specifically designed for the treatment of COVID-19. The preclinical data for EDP-235 were presented in a poster titled “EDP-235, A Potential Oral, Once-Daily Antiviral Treatment and Preventative for COVID-19,” during the International Society for Influenza and Other Respiratory Virus Diseases (ISIRV)–World Health Organization (WHO) Virtual Conference 2021. In a biochemical assay, EDP-235 inhibited the SARS-CoV-2 3CLpro protease with an IC50 of 5.8 nM. Importantly, this activity was retained against proteases from SARS-CoV-2 variants. EDP-235 potently blocked the replication of SARS-CoV-2 in multiple cellular models, including primary human airway epithelial cells, where an EC90 of 33 nM was observed. Additionally, EDP-235 was shown to have potent antiviral activity across other human coronaviruses. Mutations in the spike protein aren’t expected to significantly affect the activity of EDP-235. Compared to preclinical data from other direct-acting antivirals in development for COVID-19 today, EDP-235 appears to be among the most potent against SARS-CoV-2 in cellular assays. EDP-235 showed good human Caco-2 cell permeability and a low plasma clearance in human liver microsomes. Consistent with this in vitro data, EDP-235 had robust plasma exposure with an oral bioavailability of 95% in rats. Moreover, EDP-235 had favorable in vivo penetration into multiple target tissues, including lung, kidney, liver, and heart. These results indicate that EDP-235 has good oral bioavailability and target tissue distribution compared to other antivirals in development for SARS-CoV-2 today. Based on allometric scaling, EDP-235 is projected to have a long half-life of 16 hours with an efficacious dose of 100 to 500 mg once daily in humans. Taken together, these data indicate that EDP-235 has the potential for once-daily oral dosing with a low pill burden. Enanta has completed IND-enabling preclinical studies of EDP-235 and plans to advance the candidate into the clinic in early 2022 (27).
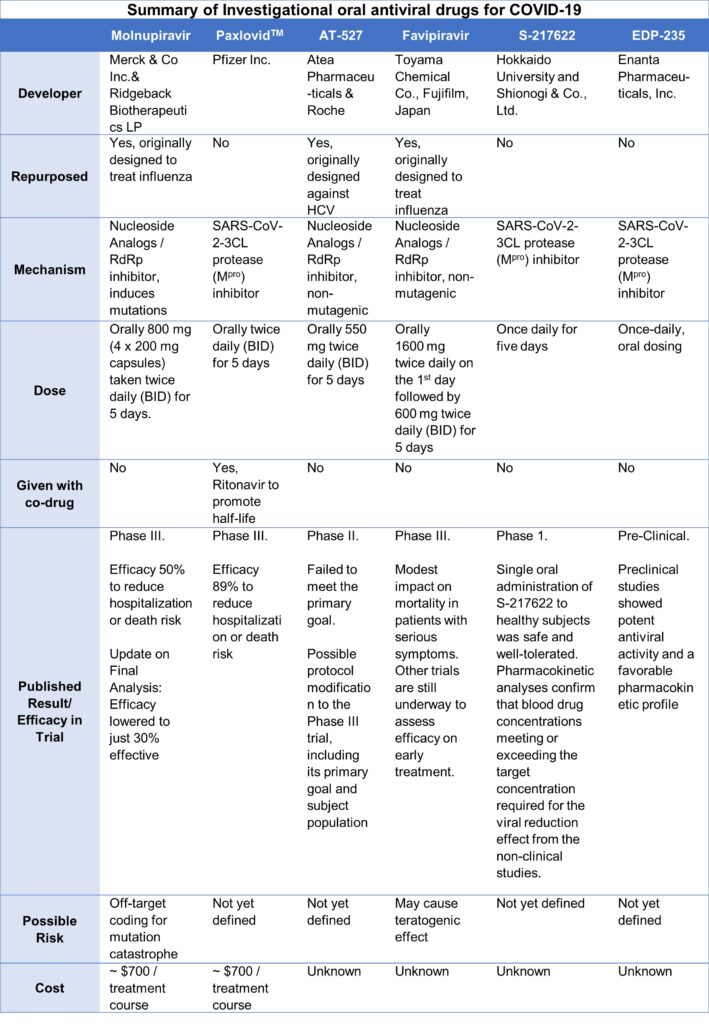
All this emerging evidence has brought new hope of a turning point in the pandemic: a not-too-distant future when a simple pill could keep infected people from dying or falling severely ill. However, will new covid treatments be elusive for developing countries, or will these treatments remain largely with nations able to pay for early access, as they have done with vaccines? It requires coordinated global action, grounded in full transparency in the procurement and delivery process as a commitment for equity and defeating the pandemic together. The summary of promising oral drugs is presented in the table below.
Reference
1. Riva L, Yuan S, Yin X, Martin-Sancho L, Matsunaga N, Pache L, et al. Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature [Internet]. 2020;586(7827):113–9. Available from: https://doi.org/10.1038/s41586-020-2577-1
2. Andersen PI, Ianevski A, Lysvand H, Vitkauskiene A, Oksenych V, Bjørås M, et al. Discovery and development of safe-in-man broad-spectrum antiviral agents. Int J Infect Dis [Internet]. 2020;93:268–76. Available from: https://www.sciencedirect.com/science/article/pii/S120197122030076X
3. Martinez MA. Lack of Effectiveness of Repurposed Drugs for COVID-19 Treatment [Internet]. Vol. 12, Frontiers in Immunology . 2021. p. 653. Available from: https://www.frontiersin.org/article/10.3389/fimmu.2021.635371
4. Food & Drug Administration – FDA. Veklury (remdesivir) EUA Letter of Approval [Internet]. 2020 [cited 2021 Nov 10]. p. 1–7. Available from: https://www.fda.gov/media/137564/download
5. Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the Treatment of Covid-19 — Final Report. N Engl J Med [Internet]. 2020 May 22;383(19):1813–26. Available from: https://doi.org/10.1056/NEJMoa2007764
6. WHO Solidarity Trial Consortium. Repurposed Antiviral Drugs for Covid-19 — Interim WHO Solidarity Trial Results. N Engl J Med [Internet]. 2020 Dec 2;384(6):497–511. Available from: https://doi.org/10.1056/NEJMoa2023184
7. Du L, Yang Y, Zhang X. Neutralizing antibodies for the prevention and treatment of COVID-19. Cell Mol Immunol [Internet]. 2021;18(10):2293–306. Available from: https://doi.org/10.1038/s41423-021-00752-2
8. Gupta A, Gonzalez-Rojas Y, Juarez E, Crespo Casal M, Moya J, Falci DR, et al. Early Treatment for Covid-19 with SARS-CoV-2 Neutralizing Antibody Sotrovimab. N Engl J Med [Internet]. 2021 Oct 27; Available from: https://doi.org/10.1056/NEJMoa2107934
9. Food & Drug Administration – FDA. Coronavirus (Covid-19) update: Fda authorizes monoclonal antibodies for treatment of covid-19 [Internet]. Vol. 13, Molecular and Cellular Pharmacology. 2021 [cited 2021 Nov 10]. p. 1–4. Available from: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-monoclonal-antibodies-treatment-covid-19
10. Kumar S, Çalışkan DM, Janowski J, Faist A, Conrad BCG, Lange J, et al. Beyond Vaccines: Clinical Status of Prospective COVID-19 Therapeutics [Internet]. Vol. 12, Frontiers in Immunology . 2021. p. 4064. Available from: https://www.frontiersin.org/article/10.3389/fimmu.2021.752227
11. Willyard C. How antiviral pill molnupiravir shot ahead in the COVID drug hunt. Nat. 2021;
12. V’kovski P, Kratzel A, Steiner S, Stalder H, Thiel V. Coronavirus biology and replication: implications for SARS-CoV-2. Nat Rev Microbiol [Internet]. 2021;19(3):155–70. Available from: https://doi.org/10.1038/s41579-020-00468-6
13. Cannalire R, Cerchia C, Beccari AR, Di Leva FS, Summa V. Targeting SARS-CoV-2 Proteases and Polymerase for COVID-19 Treatment: State of the Art and Future Opportunities. J Med Chem [Internet]. 2020 Nov 13; Available from: https://doi.org/10.1021/acs.jmedchem.0c01140
14. Jang WD, Jeon S, Kim S, Lee SY. Drugs repurposed for COVID-19 by virtual screening of 6,218 drugs and cell-based assay. Proc Natl Acad Sci. 2021;118(30).
15. Williamson BN, Feldmann F, Schwarz B, Meade-White K, Porter DP, Schulz J, et al. Clinical benefit of remdesivir in rhesus macaques infected with SARS-CoV-2. Nature [Internet]. 2020;585(7824):273–6. Available from: https://doi.org/10.1038/s41586-020-2423-5
16. Malone B, Campbell EA. Molnupiravir: Coding for catastrophe. Nat Struct Mol Biol. 2021;28(9):706–8.
17. MERCK. Merck and Ridgeback ’ s Investigational Oral Antiviral Molnupiravir Reduced the Risk of Hospitalization or Death by Approximately 50 Percent Compared to Placebo for Patients with Mild or Moderate COVID-19 in Positive Interim Analysis of Phase 3 Study [Internet]. 2021 [cited 2021 Nov 10]. p. 1–6. Available from: https://www.merck.com/news/merck-and-ridgebacks-investigational-oral-antiviral-molnupiravir-reduced-the-risk-of-hospitalization-or-death-by-approximately-50-percent-compared-to-placebo-for-patients-with-mild-or-moderat/
18. Owen DR, Allerton CMN, Anderson AS, Aschenbrenner L, Avery M, Berritt S, et al. An oral SARS-CoV-2 Mpro inhibitor clinical candidate for the treatment of COVID-19. Science (80- ). 2021;eabl4784.
19. Pfizer Inc. PFIZER’S NOVEL COVID-19 ORAL ANTIVIRAL TREATMENT CANDIDATE REDUCED RISK OF HOSPITALIZATION OR DEATH BY 89% IN INTERIM ANALYSIS OF PHASE 2/3 EPIC-HR STUDY [Internet]. 2021 [cited 2021 Nov 10]. Available from: https://www.pfizer.com/news/press-release/press-release-detail/pfizers-novel-covid-19-oral-antiviral-treatment-candidate
20. Good SS, Westover J, Jung KH, Zhou X-J, Moussa A, La Colla P, et al. AT-527, a double prodrug of a guanosine nucleotide analog, is a potent inhibitor of SARS-CoV-2 in vitro and a promising oral antiviral for treatment of COVID-19. Antimicrob Agents Chemother. 2021;65(4):e02479-20.
21. Atea Pharmaceuticals. Atea Pharmaceuticals Provides Update and Topline Results for Phase 2 MOONSONG Trial Evaluating AT-527 in the Outpatient Setting [Internet]. 2021 [cited 2021 Nov 10]. p. 7–9. Available from: https://ir.ateapharma.com/news-releases/news-release-details/atea-pharmaceuticals-provides-update-and-topline-results-phase-2#:~:text=PDF Version-,Atea Pharmaceuticals Provides Update and Topline Results for Phase 2,527 in the Outpatient Setting&text=“We remain committed to our,-19 continues to evolve.”
22. Allen CNS, Arjona SP, Santerre M, Sawaya BE. Potential use of RNA-dependent RNA polymerase (RdRp) inhibitors against SARS-CoV2 infection. All Life. 2020;13(1):608–14.
23. Furuta Y, Komeno T, Nakamura T. Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc Jpn Acad Ser B Phys Biol Sci [Internet]. 2017;93(7):449–63. Available from: https://pubmed.ncbi.nlm.nih.gov/28769016
24. Naesens L, Guddat LW, Keough DT, van Kuilenburg ABP, Meijer J, Vande Voorde J, et al. Role of human hypoxanthine guanine phosphoribosyltransferase in activation of the antiviral agent T-705 (favipiravir). Mol Pharmacol. 2013 Oct;84(4):615–29.
25. Hassanipour S, Arab-Zozani M, Amani B, Heidarzad F, Fathalipour M, Martinez-de-Hoyo R. The efficacy and safety of Favipiravir in treatment of COVID-19: a systematic review and meta-analysis of clinical trials. Sci Rep [Internet]. 2021;11(1):11022. Available from: https://doi.org/10.1038/s41598-021-90551-6
26. Shionogi. Notice Regarding the Initiation of a Phase 2/3 Clinical Trial for a COVID-19 Therapeutic Agent in Japan [Internet]. 2011 [cited 2021 Nov 10]. p. 32–3. Available from: https://www.shionogi.com/global/en/news/2021/09/e-210928.html 27. Enanta Pharmaceutical. Enanta Pharmaceuticals Presents New Data for EDP- 235 , its Lead Oral Protease Inhibitor Designed for the Treatment of COVID-19 , at the ISIRV – WHO Virtual Conference 2021 [Internet]. 2021 [cited 2021 Nov 10]. p. 2021–3. Available from: https://www.enanta.com/research/COVID-19/default.aspx
27. Enanta Pharmaceutical. Enanta Pharmaceuticals Presents New Data for EDP- 235 , its Lead Oral Protease Inhibitor Designed for the Treatment of COVID-19 , at the ISIRV – WHO Virtual Conference 2021 [Internet]. 2021 [cited 2021 Nov 10]. p. 2021–3. Available from: https://www.enanta.com/research/COVID-19/default.aspx
Leave a reply
Most Commented