HEPATITIS OF UNKNOWN ORIGIN IN CHILDREN – WHAT WE KNOW, AND WHAT WE DON’T KNOW (YET) FOR NOW
By: Yan Mardian
- Event Background
On 31 March 2022, Public Health Scotland was alerted to five children aged 3–5 years at Glasgow children’s hospital with severe hepatitis of unknown etiology within three weeks. This cluster exceeded the expected number of cases of hepatitis of unknown etiology, fewer than four per year. The United Kingdom (UK) informed these cases to the World Health Organization’s International Health Regulations (IHR) notification system on 5 April 2022 (testing had excluded viral hepatitis types A, B, C, D, and E and other known causes of acute hepatitis). Following this alert, the USA and several European Union, European Economic Area (EU/EEA), and other countries reported suspected cases. As of 3 May 2022, there have been 163 cases of acute non-A-E hepatitis with serum transaminases greater than 500 IU/l identified in children under 16 years old in the UK since 1 January 2022. This resulted from an active case-finding investigation commencing in April, which identified retrospective and prospective cases.
As of 21 April 2022, at least 11 countries in the WHO European Region and one country in the WHO Region of the Americas have reported the cases. Cases are aged one month to 16 years old. Seventeen children (approximately 10%) required liver transplantation; at least one death was reported. In November 2021, clinicians at a large children’s hospital in Alabama, US, notified the Centers for Disease Control and Prevention (CDC) of five pediatric patients with significant liver injury, including three with acute liver failure. All five patients had tested positive by blood PCR for adenovirus, and all were previously healthy. None had COVID-19 or common hepatitis viruses detected on testing. Case-finding efforts at this hospital identified four additional pediatric patients with hepatitis, all with adenovirus DNA identified in the blood by PCR, for a total of nine patients admitted during the five months from October 2021 through February 2022. A multi-disciplinary expert team reviewed clinical and epidemiological data from the first five children: vomiting in preceding weeks, jaundice, and exceptionally high levels of transaminases, often greater than 2000 international units per liter (IU/L; n.v. <40 IU/L), were the main features.

- World Health Organization (WHO) and Joint Euro-pean Centre for Disease Prevention and Control (ECDC) Working Case Definition
a. Confirmed: N/A at present
b. Probable: A person presenting with acute hepatitis* with serum transaminase >500 IU/L (AST or ALT), who is 16 years and younger, since 1 October 2021.
c. Epi-linked: A person presenting with acute hepatitis* of any age who has had close contact with a probable case, since 1 October 2021.
*(non-hep A-E (If hepatitis A-E serology results are await-ed, but other criteria are met, can be reported, and will be classified as “pending classification.” Cases with other explanations for their clinical presentation are discarded))
- Case definition used by the European Union, European Economic Area (EU/EEA)
a. Confirmed: A person presenting with acute hepatitis* with serum transaminase >500 IU/l (Aspartate Trans-aminase-AST or Alanine Transaminase-ALT), who is 10 years and under, since 1 January 2022.
b. Possible: A person presenting with acute hepatitis* with serum transaminase >500 IU/l (AST or ALT), who is 11 to 16 years, since 1 January 2022.
c. Epi-linked: A person presenting with acute hepatitis* of any age who has had close contact with a confirmed case, since 1 January 2022.
*(non hep A-E or an expected presentation of metabolic, inherited or genetic, congenital or mechanical cause*) **Confirmed and possible cases should be reported based on the clinical judgment if some hepatitis A-E virus results are awaited, or if there is an acute on chronic hepatic presentation with a metabolic, inherited or genetic, con-genital, mechanical, or other underlying cause. If hepatitis A-E serology results are awaited, but other criteria are met, these will be classified as ‘pending classification.’
- What Have We Learned, So Far?
- Epidemiology and Clinical Features
Hepatitis is a condition characterized by the inflammation of the hepatic parenchyma. The inflammation may be acute, lasting typically less than six months with subsequent normalization of liver function, or it may be chronic. Non-infectious causes of hepatitis in children include immunologic conditions (e.g., autoimmune diseases), metabolic diseases (e.g., Wilson’s disease, tyrosinemia), and exposure to toxins or drugs (e.g., acetaminophen). The most common infectious agents are the primary hepato-tropic viruses (Hepatitis A, B, C, D, E). Other viruses that may cause acute hepatitis include Epstein-Barr virus (EBV), cytomegalovirus (CMV), parvovirus, enteroviruses, adenoviruses, rubella virus, herpesviruses (HHV-1, HHV-2, HHV-6, HHV-7) and human immunodeficiency virus (HIV). Other infectious agents include Brucella spp, Coxiella burnetii, and Leptospira.
Common symptoms of acute hepatitis (myalgia, nausea, vomiting, lethargy, fatigue, fever, abdominal pain, and diarrhea) sometimes persist for several weeks. A high proportion of acute infections with the hepatitis viruses are asymptomatic. For hepatitis A and B, the infection is much more likely to produce a minor or asymptomatic illness among children than among adults. Jaundice is commonly associated with acute hepatitis but may not show in many viral hepatitis cases. Death from acute viral hepatitis is rare and usually results from the development of fulminant hepatitis, acute liver failure (ALF) with hepatic encephalopathy. The risk of ALF resulting from fulminant viral hepatitis is associated with increasing age and pre-existing liver disease. Impaired coagulation with a prolonged prothrombin time is one of the classic markers of ALF. Hepatic encephalopathy can be subtle, especially in infants. Bone marrow failure occurs in a few children with ALF, ranging from mild pancytopenia to aplastic anemia. Without liver transplantation, mortality in children with ALF is very high. In up to 50% of ALF cases in children, the cause cannot be identified, and they are classified as indeterminate. The treatment of indeterminate ALF cases is general supportive measures and liver transplantation.
As of 3 May, there were 118 cases of hepatitis of unknown origin in children in England. No known epidemiologically linked the cases in England. Forty potential cases in England are awaiting classification pending further data. Cases are predominantly aged between 3 and 5 years old (66, 56.9%), a median age of 3 (interquartile range 3 to 4 years), and 50% are female. The majority are of white ethnicity (92 out of 107, 86.0%) where information was available. Many cases had gastrointestinal symptoms in the weeks preceding the onset of jaundice. Of the 118 cases investigated in England as of 3 May, the most common presentation reported in cases remains jaundice (84 out of 118, 71.2%), followed by vomiting (74 out of 118, 62.7%). Pale stools were also frequently reported (50.0%). Gastrointestinal symptoms were commonly reported at presentation, including diarrhea. (44.9%), nausea (30.5%) and abdominal pain (41.5%). Additionally, lethargy (50.0%), fever (30.5%) and less frequently, respiratory symptoms (18.6%) were reported. These clinical findings are consistent with those described among cases reported by Scotland, although none of the Scottish cases were reported to have had a fever. Alt-hough all cases had high transaminase levels in line with the case definition, most of the children reported from Scotland had transaminases over 2000 IU/L. Eleven cases have received a liver transplant. No cases resident in the UK has died.
Nine patients with hepatitis of unknown etiology at Children’s of Alabama in Alabama, US, were recorded from October 2021 to February 2022. These patients were from geographically distinct parts of the state; no epidemiologic links among patients were identified. The median age at admission was two years, 11 months (IQR = 1 year, 8 months to 5 years, nine months), and seven patients were female. All patients were immunocompetent with no clinically significant medical comorbidities. Before admission, seven, six, and three patients reported vomiting, diarrhea, and upper respiratory symptoms, respectively. Eight patients had scleral icterus at admission, seven had hepatomegaly, six had jaundice, and one had encephalopathy. Elevated transaminases were detected among all patients§ (alanine aminotransferase [ALT] range = 603–4,696 U/L; aspartate aminotransferase [AST] range = 447–4,000 U/L); total bilirubin ranged from normal to elevated (range = 0.23–13.5 mg/dL, elevated in eight patients). Three patients developed acute liver failure. Two of them were treated with cidofovir (off-label use) and steroids and were transferred to a different medical facility where they underwent liver transplantation. All patients have recovered or are recovering, including the two transplant recipients.
In Israel, 12 cases came in the last four months; five were hospitalized in Shaare Zedek Medical Center in Jerusalem and seven in Schneider Children’s Medical Center in Petah Tikva. Two of the children in Schneider suffered from liver failure, prompting doctors to carry out liver transplants. The condition of the other children improved quickly after treatment with steroids, and they were released from the hospitals. 11 out of the 12 children were infected with the coronavirus in the last year.
- Histopathological examinations
In addition to local assessments, an additional review of all available liver samples was undertaken by a single expert histopathologist. These specimens included six ex-planted (removed) livers and eight biopsies from a combination of English and Scottish cases. The specimens demonstrated variable severity ranging from mild hepatocellular injury to massive hepatic necrosis. The overall pattern seen is non-specific, and there is no clear identifiable cause from the histopathology results. On hematoxylin and eosin (H and E) staining, the inflammatory response was variable throughout the specimens reviewed. Further immunohistochemistry for lymphocytic subpopulations is planned. Adenovirus immunohistochemistry has been reported from 9 of the 14 samples and shown immunoreactivity in the intrasinusoidal lumen but not in residual hepatocytes. This is likely a non-specific finding. One case underwent adenovirus PCR of liver tissue which was negative. Liver biopsies from six Alabama patients demonstrated various degrees of hepatitis with no viral inclusions observed, no immuno-histochemical evidence of adenovirus, or no viral particles identified by electron microscopy.
- Microbiological
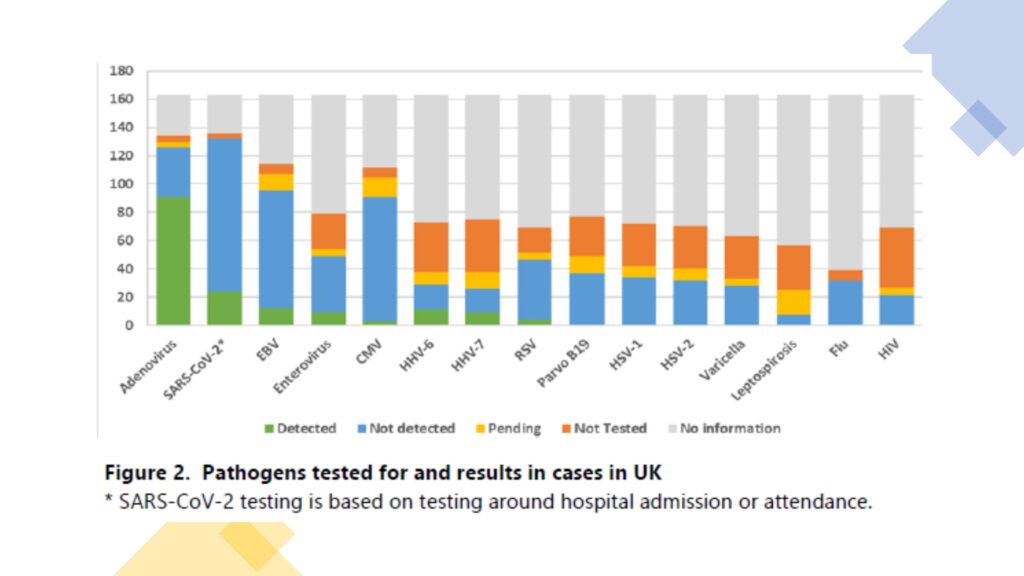
Most testing information available from cases reported to date is from England. However, not all cases have been tested for the same set of pathogens, at or around the time of admission. Adenovirus remains the most frequently detected potential pathogen. Amongst 163 UK cases, 126 have been tested for adenovirus of which 91 had adenovirus detected (72%). Amongst the cases, adenovirus has primarily been detected in the blood. Further analysis relates to cases from England, of which adenovirus was detected in 67 of 89 cases that have been tested. Of the 8 England-resident patients who required a liver transplant, seven were tested for adenovirus in blood samples and the virus was detected in all 7. Of the 22 cases where adenovirus was not detected, 6 had not had testing on blood which appears to be the most relevant sample type for the syndrome, 5 were tested on plasma not whole blood, and a further 9 of unknown sample types had been tested in a hospital laboratory but not retested by the reference laboratory, although we are aware of potential performance differences between as-says in clinical use. It is therefore not possible to definitively rule out adenovirus in these cases.
Typing by partial hexon gene sequencing consistently shows that the adenovirus present in the blood is type 41F (18 of 18 cases with an available result). Whole-genome sequencing (WGS) has been attempted on multiple samples from cases, but the low viral load in blood samples and limited clinical material from historic cases mean that it has not been possible to get a good quality full adenovirus genome from a case as yet.
SARS-CoV-2 has been detected in 24 cases of 132 with available results (18%). Five cases with a positive test result for SARS-CoV-2 also have associated variant information from WGS. All 5 sequences are classified as VOC-22JAN-01 (lineage BA.2). Four of the 5 sequences contain mutations in addition to those expected to be present in all BA.2 sequences, but the mutations do not occur in more than one sequence. Two of the 6 sequences are from the time of hepatitis presentation, the remaining 4 sequences are from 3-, 8-, and 15-days post hepatitis presentation. Serological testing is in the process to explore prior infection further, however, the high population cumulative prevalence of SARS-CoV-2 will make the interpretation of this data challenging. Four cases were co-infected with adenovirus and SARS-CoV-2. A range of other possible pathogens have been detected in a low proportion of cases and are of uncertain significance. However, the inclusive nature of the UKHSA case definition intentionally will pick up some cases of non-A-E hepatitis with recognized causes.

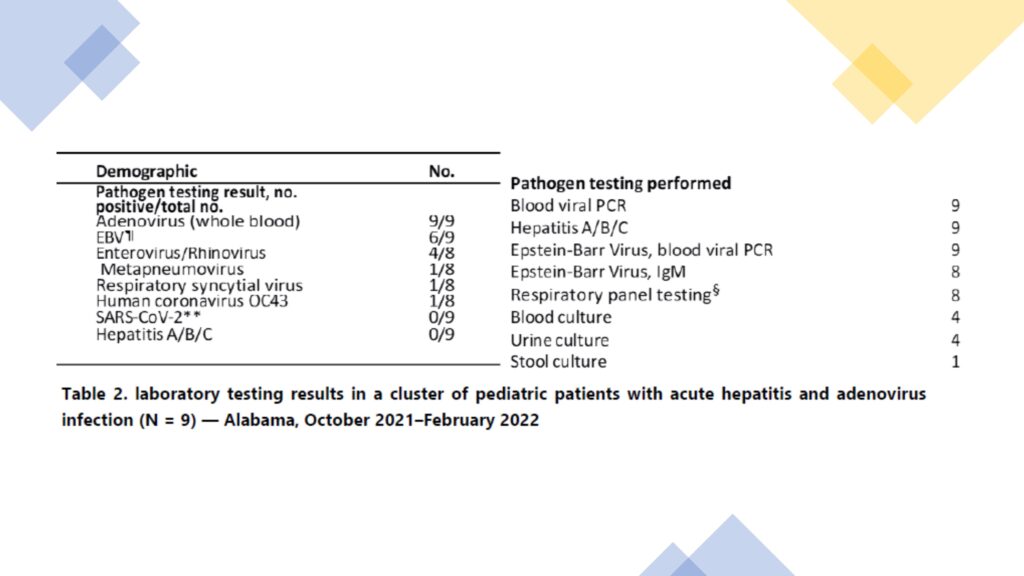
In US cases, Adenovirus was detected in whole blood specimens from all patients by real-time PCR testing (initial viral load range = 991–70,680 copies/mL). Hexon gene hypervariable region sequencing was performed on specimens from five patients, and adenovirus type 41 was detected in all five specimens. Plasma specimens from two patients who underwent liver transplantation were negative for adenovirus by real-time PCR testing upon arrival at the receiving medical facility, but both patients received positive test results when retested by the same real-time PCR test using a whole blood specimen. Low viral loads precluded sequencing among three patients, and residual specimens were not available for sequencing for one patient. Seven patients were coinfected with other viral pathogens. Six received positive test results for Epstein-Barr virus (EBV) by PCR testing but negative test results for EBV immunoglobulin M (IgM) antibodies (one patient did not have IgM testing), suggesting that these were likely not acute infections but rather low-level reactivation of previous infections. Other detected viruses included enterovirus/rhinovirus, metapneumovirus, respiratory syncytial virus, and human coronavirus OC43. All patients received negative test results for hepatitis viruses A, B, and C. Several other causes of pediatric hepatitis and infections were ruled out, including autoimmune hepatitis, Wilson dis-ease, bacteremia, urinary tract infections, and SARS-CoV-2 infection. None of the children had documented history of previous SARS-CoV-2 infection.
- Metagenomics
Amongst the UK cases, metagenomics has been performed on 19 samples: 14 samples from 11 English cases (6 blood, 4 liver, 2 serum, and 2 EDTA-Plasma) and 5 Scottish samples from 5 Scottish cases (all sera). Metagenomics undertaken on blood and liver tissue has detected primarily adeno-associated virus 2 (AAV-2) in high quantities. Whilst contamination was originally suspected, AAV-2 is now detected in multiple samples from different hospital sources and tested in more than one sequencing laboratory. This finding is of uncertain significance and may represent a normal reactivation of AAV-2 during acute viral infection (for example, adenovirus) or during liver injury of another cause. It is not unusual to detect bystanders, reactivating, or other incidental species during metagenomic sequencing. However, given the presence of AAV-2 in a number of cases, the significance will be further explored through the testing of additional sets of controls.
AAV2 is a dependoparvovirus that is typically dependent on other viruses including adenovirus and herpesviruses to replicate. Non-pathogenic human infection is common and latent viruses may reactivate in some circumstances, again with no clinical consequence. The hypotheses which are under consideration to explain the detection of AAV2 in metagenomic data are:
a. Upregulation of AAV2 due to adenovirus or another acute viral infection.
b. Upregulation of AAV2 due to liver injury.
c. Contamination (for example of a reagent). Laboratory contamination is now considered less likely given that AAV2 has been detected in 2 testing laboratories with the appropriate negative assay controls but remains a possible explanation.
d. Undetermined role in the pathogenesis of the syn-drome.
- Toxicology
Toxicological investigations continue with no positive findings to date. Detection of paracetamol is likely to be related to appropriate therapeutic use (also noted in the trawling questionnaires) which would not be a concern, however, verification work is being undertaken to confirm this.
- Host investigations
Host (for example, immunological) investigations require full research consent and are undertaken under the Inter-national Severe Acute Respiratory and Emerging Infection Consortium (ISARIC) Clinical Characterization Protocol. Thirty-seven cases have been recruited to the ISARIC clinical characterization protocol to date and retrospective and prospective recruitment continues.
- Possible Exposures
Investigations have included interviews of parents conducted by public health specialists to assess a broad range of different exposures (trawling questionnaires). In the first 60 case patients in England with data available, no notable features or common exposures were observed in travel, family structure, parental occupation, diet, water source, or potential exposures to toxicants, and no association with prior immunosuppression. The public health agencies of Wales, Scotland and Northern Ireland report similar findings through their investigations. Public Health Scotland also report that there are 2 pairs of epidemiologically linked cases.
A review of UK trawling questionnaire responses has found relatively high numbers of dog-owning families or other dog exposures in cases (64 of 92 where data was available, 70%). The significance of this finding is being explored. Pet dog ownership is common in the UK. There are limited data on background rates of pet ownership in families of young children, non-household dog contact reporting may include transient non-significant contact, and the nature of trawling questionnaire investigations means that some responses may be high through the play of chance due to the large numbers of questions asked.
Approximately three-quarters of respondents in data for England mentioned paracetamol use. Fewer reported ibuprofen use and none reported aspirin use. While paracetamol is an important hepatotoxic agent in overdose, there have been no reports of paracetamol hepatoxic presentations or histories from any of the clinical units. The prevalence of paracetamol use is considered consistent with the guidance on the management of acute illness in children.
COVID-19 vaccinations are not recommended by the Joint Committee on Vaccination and Immunisation for children aged under 5. They are available for children aged 5 and over. There were no COVID-19 vaccinations recorded in cases aged under 5, the age group which makes up over 75% of hepatitis cases. There are fewer than 5 older case patients recorded as having had a COVID-19 vaccination prior to hepatitis onset. There is no evidence of a link between COVID-19 vaccination and acute hepatic syndrome.
- Working Hypothesis
The following hypotheses are all being actively tested by the investigations in process. There are increased pediatric acute non-A-E hepatitis presentations due to:
I. A normal adenovirus infection, due to one of:
a) Abnormal susceptibility or host response which allows adenovirus infection to progress more frequently to hepatitis (whether direct or immuno-pathological), for example from lack of exposure during the coronavirus (COVID-19) pandemic.
b) An exceptionally large wave of normal adenovirus infections, causing a very rare or under-recognised complication to present more frequently.
c) Abnormal susceptibility or host response to adenovirus due to priming by a prior infection with SARS-CoV-2 (including Omicron restricted) or another infection.
d) Abnormal susceptibility or host response to adenovirus due to a coinfection with SARS-CoV-2 or another infection.
e) Abnormal susceptibility or host response to adenovirus due to a toxin, drug or environmental exposure.
II. A novel variant adenovirus, with or without a contribution from a cofactor as listed above.
III. A post-infectious SARS-CoV-2 syndrome (including an Omicron restricted effect).
IV. A drug, toxin or environmental exposure.
V. A novel pathogen either acting alone or as a coinfection.
VI. A new variant of SARS-CoV-2
- Adenovirus: Is He Guilty?
1) Virological features and clinical presentation
Adenoviruses are a group of double-stranded DNA non-enveloped DNA (dsDNA) viruses belonging to the genus Mastadenovirus of the Adenoviridae family. Adenovirus genomes share a central conserved part that can be used for detection purposes. Human adenoviruses (HAdV) are separated into seven genetically distinguishable species (HAdV-A through HAdV-G) and are currently classified into more than 100 genotypes and 52 serologically distinct types. Species A, B, C, D, E, and F circulate globally and have been implicated in outbreaks of infection in humans. Different genome types (or genomic variants) can be distinguished within the same serotype by re-striction enzyme analysis of genomic DNA. Different types display different tissue tropisms, which may correlate with clinical manifestation and may circulate at a given time in different countries or regions causing trans-mission of novel strains between countries or across continents and replacement of dominant viruses with new strains.

The incubation period for respiratory adenoviruses is estimated to range between two and 14 days and for enteric ones between three and 10 days. The incidence of adenovirus infection peaks between the ages of six months and five years, but the highest incidences have been described among children under two years. The most common clinical features are keratoconjunctivitis (HAdV types 5, 8, 19, and 37), acute respiratory symptoms (HAdV types 1-5, 7, 14, and 21), urethritis in men by types 8 and 37, or gastroenteritis (HAdV-types 31, 40 and 41). More rare manifestations include kidney disease, hemorrhagic cystitis, or hepatitis. Adenovirus (HAdV-40 and HAdV-41) is considered one of the most important causative agents of acute viral gastroenteritis in young children. Although HAdV infections are generally self-limiting in healthy children; immunocompromised individuals, for example acute leukemia patients, bowel transplant patients, and stem cell and solid-organ transplant recipients, are at higher risk for developing severe and disseminated disease. Acute liver failure from adenovirus is rare and is described especially in immuno-compromised patients. Depending on the species, these viruses may infect respiratory, conjunctival, gastrointestinal, and genitourinary sites. To note, fulminant hepatitis is a rare complication of adenoviral infection. Latent infection with HAdVs may occur with the virus residing in renal, lymphoid, or other tissues for many years, with re-activation sometimes occurring in severely immunosuppressed individuals.
2) Circulation
Adenoviruses circulate throughout the year. In the USA, the highest numbers of detections of adenoviruses associated with conjunctivitis in a 30-year study period have been from July to September and the lowest from April to June every year. Higher circulation of adenoviruses has been detected in Brazil from April-May and July to October and in China, a higher prevalence peaked in April and October. Uncertainties remain about the seasonality of adenovirus in the EU/EEA and whether it is type-specific.
3) Routes transmission
Transmission can occur by direct contact with infected individuals through inhalation of droplets, faecal-oral route, and conjunctival inoculation, or indirectly through exposure to contaminated objects (fomites). Infections may spread rapidly among closed populations, for example in hospitals, schools and nurseries, and se-vere outbreaks of respiratory infection or keratoconjunctivitis due to HAdV have been described linked to a variety of virus types. Some outbreaks of more severe disease have been reported among groups of immunocompromised people.
4) Role of Adenovirus in the landscape of viral hepatitis in children?
Adenovirus type 41, the apparently implicated adenovirus type, typically presents as diarrhea, vomiting, and fever, often accompanied by respiratory symptoms. While there have been case reports of hepatitis in immunocompromised children with adenovirus infection, adenovirus type 41 is not known to be a cause of hepatitis in otherwise healthy children. Reports of positive adenovirus tests from any site in 1- to 4-year-olds are higher compared to the previous 5 years. Between November 2021 to April 2022, approximately 200 to 300 cases of adenovirus were reported into SGSS per week compared to 50 to 150 cases per week in the pre-pandemic period and less than 50 cases per week between March 2020 and May 2021. The increase in younger age groups began in November 2021.
In a recent systematic review of the global epidemiology of viral-induced acute liver failure, the burden of acute liver failure after infection with hepatitis B virus, hepatitis A virus, hepatitis C virus, hepatitis E virus, herpes simplex virus/human herpesvirus, cytomegalovirus, Epstein-Barr virus, and parvovirus B19 was estimated. The prevalence of hepatitis A-induced acute liver failure was markedly lower in countries with routine hepatitis A immunization versus no routine hepatitis A immunization. Hepatitis E virus was the most common etiological cause of viral-induced acute liver failure reported in this review. In addition, viral-induced acute liver failure had poor outcomes as indicated by high fatality rates, which appear to increase with poor economic status of the studied countries. Unfortunately, data were largely missing for acute liver failure after infection with varicella-zoster virus, human parainfluenza viruses, yellow fever virus, coxsackievirus, and/or Adenovirus.
Further investigative work, including WGS of multiple cases, is required before any firm conclusions can be drawn on the characterization of the adenoviruses involved. Adenovirus WGS as well as metagenomic sequencing have commenced on case samples. The low levels of adenovirus present in the blood are challenging for the recovery of high-quality genomes. For blood samples with attempted WGS, cycle threshold values range from 32 to 37. There are currently very limited whole-genome adenovirus sequence data available in the public domain, particularly for enteric adenoviruses. Academic and clinical centers which have or can generate adenovirus WGS data are asked to share consensus genomes to an International Nucleotide Sequence Database Collaboration such as GenBank to assist characterization of circulating adenovirus strains internationally.

5) Hypothesis of AdV potentiated SARS-CoV-2 superantigen-mediated pathology
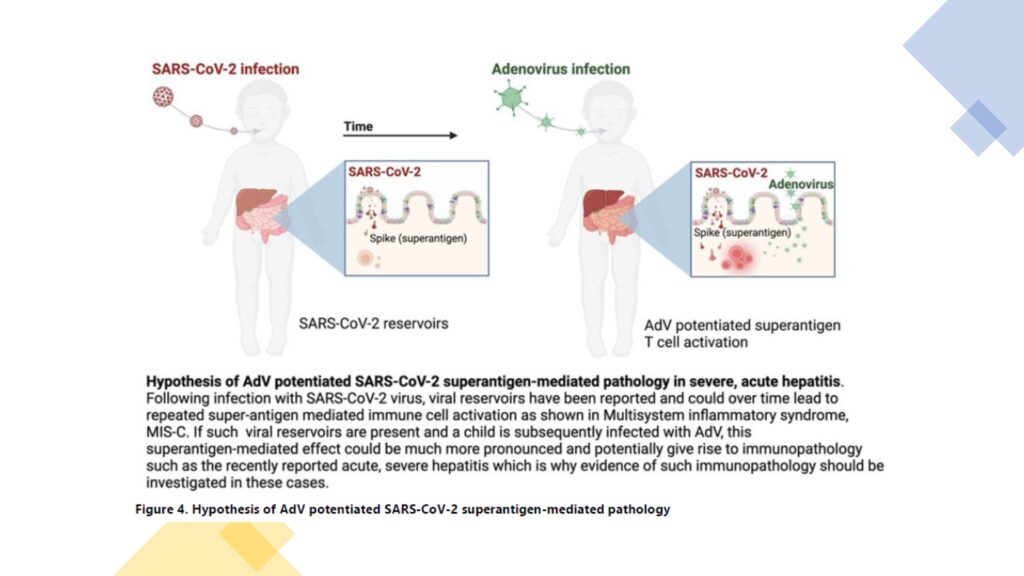
The SARS-CoV-2 has been identified in 18% of reported cases in the UK and 11 (11%) of 97 cases in England with available data tested SARS-CoV-2 positive on admission; a further three cases had tested positive within the 8 weeks prior to admission. Ongoing serological testing is likely to yield greater numbers of children with severe acute hepatitis and previous or current SARS-CoV-2 infection. Eleven of 12 Israeli patients were reported to have had COVID-19 in recent months, and most reported cases of hepatitis were in patients too young to be eligible for COVID-19 vaccinations. SARS-CoV-2 infection can result in viral reservoir formation. SARS-CoV-2 viral persistence in the gastrointestinal tract can lead to a repeated release of viral proteins across the intestinal epithelium, giving rise to immune activation. Such repeated immune activation might be mediated by a superantigen motif within the SARS-CoV-2 spike protein that bears resemblance to Staphylococcal enterotoxin B, triggering broad and non-specific T-cell activation. This superantigen mediated immune-cell activation has been proposed as a causal mechanism of the multisystem inflammatory syndrome in children.
Acute hepatitis has been reported in children with multi-system inflammatory syndrome, but coinfection of other viruses was not investigated. There is a hypothesis that the recently reported cases of severe acute hepatitis in children could be a consequence of adenovirus infection with intestinal tropism in children previously infected by SARS-CoV-2 and carrying viral reservoirs. In mice, adenovirus infection sensitises to subsequent Staphylococcal-enterotoxin-B-mediated toxic shock, leading to liver failure and death. This outcome was explained by adenovirus-induced type-1 immune skewing, which, upon subsequent Staphylococcal enterotoxin B administration, led to excessive IFN-γ production and IFN-γ-mediated apoptosis of hepatocytes. Translated to the current situation, it is suggested that children with acute hepatitis be investigated for SARS-CoV-2 persistence in stool, T-cell receptor skewing, and IFN-γ upregulation because this could provide evidence of a SARS-CoV-2 superantigen mechanism in an adenovirus-41F-sensitised host. If evidence of superantigen mediated immune activation is found, immunomodulatory therapies should be considered in children with severe acute hepatitis.
- Current guideline for testing
In addition to case findings, when testing probable and epidemiologically linked cases, appropriate samples should be collected to perform the tests outlined in Table 4. ECDC recommends the early collection of multiple specimen types from the cases under investigation and testing with different diagnostic methods for prompt detection of possible causative agents. Countries should include adenovirus testing for children with severe acute hepatitis, at the same time as testing for hepatitis A-E. Preliminary data indicate that whole blood is an important sample matrix to test for viruses. It will be important to store specimens (e.g., serum and EDTA blood, nasopharyngeal/throat swabs (for bacterial and viral testing), fecal, and urine specimens) for possible further diagnostic testing and typing as required. Adenovirus and/or SARS-CoV-2 positive samples should be typed.
If diagnostics are not available locally, then specimens should be referred to national laboratories, including for typing and pathogen characterization. Quantification of positive PCR findings in blood samples should be conducted with cycle threshold (Ct) value as a proxy, and if possible, using sequential sampling over a longer time period. Institutes with metagenomic capacities can consider metagenomic analyses of samples for probable and epidemiologically linked cases. Samples for potential analysis can include blood and available liver biopsies but can be extended to any relevant samples. As the etiology remains unknown, relevant toxicology and environmental studies should also be considered where possible. Laboratory screening for metabolic and autoimmune diseases is recommended to exclude other non-infectious causes.
- Situation in Indonesia
Indonesia Health Minister Budi Gunadi Sadikin in a written statement on Monday (05/10) urged the general public to be aware and take preventive measures against the threat of acute hepatitis that has been found in multiple countries across the globe and is believed to have entered Indonesia. Currently, there have been three reported cases believed to be linked to this disease claiming the lives of three children in Indonesia.
The minister added that as of May 11, there are 18 cases of suspected acute hepatitis of unknown etiology. The first three cases in Indonesia were reported on April 27, 2022, a few days after the World Health Organization (WHO) reported the outbreak in Europe. The Health Ministry quickly followed up on this incident by issuing a Circular (SE) regarding the Precautions for the Discovery of Acute Hepatitis of Unknown Aetiology. The Ministry is keeping close communication with the United States CDC and Britain’s government regarding the details of the acute hepatitis outbreak, though unfortunately, there are yet conclusive explanations on why this disease can spread wildly. Indonesia, in partnership with the WHO and the U.S. government, is currently conducting tests to uncover the cause of the outbreak.

As of May 4, the Indonesian Paediatric Society had issued several recommendations in response to this outbreak, adapting to formerly explained guidelines issued by ECDC and US-CDC. The screening and management algorithms are as follows (provided in Bahasa Indonesia).
References:
- World Health Organization (23 April 2022). Disease Out-break News; Multi-Country – Acute, severe hepatitis of unknown origin in children. Available at: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON376
- Baker, J.M.; Buchfellner, M.; Britt, W.; Sanchez, V.; Potter, J. L.; Ingram, L.A.; Shiau, H.; Sanchez, L.H.G.; Saaybi, S.; Kelly, D.; et al. Acute Hepatitis and Adenovirus Infection Among Children—Alabama, October 2021–February 2022. MMWR Morb. Mortal. Wkly. Rep. 2022. Available online: https://www.cdc.gov/mmwr/volumes/71/wr/mm7118e1.htm?s_cid=mm7118e1_w
- European Centre for Disease Prevention and Control. Increase in severe acute hepatitis cases of unknown etiology in children – 28 April 2022. ECDC: Stockholm; 2022. https://www.ecdc.europa.eu/en/publications-data/increase-severe-acute-hepatitis-cases-unknown-aetiology-children
- UK Health Security Agency. Investigation into acute hepatitis of unknown etiology in children in England: technical briefing. May 2022. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1073704/acute-hepatitis-technical-briefing-2.pdf
- Lynch III JP, Kajon AE. Adenovirus: epidemiology, global spread of novel serotypes, and advances in treatment and prevention. Semin Respir Crit Care Med. 2016;37 (04):586-602. Available at: https://www.thieme-connect.com/products/ejournals/html/10.1055/s-0036-1584923
- Centers for Disease Control and Prevention (CDC). CDC Alerts Providers to Hepatitis Cases of Unknown Origin. Atlanta: CDC; 2022. Available at: https://www.cdc.gov/media/releases/2022/s0421-hepatitis-alert.html
- Ghebremedhin, B., MD, MSc. (2014). Human adenovirus: Viral pathogen with increasing importance, European Journal of Microbiology and Immunology EuJMI, 4(1), 26-33 Retrieved May 10, 2022, from https://akjournals.com/view/journals/1886/4/1/article-p26.xml
- Kiwan P, Hamod DA (2017) Adenoviral Hepatitis in an Immunocompetent Child: Case report. J Pediatr Neonatal Care 7(3): 00290. DOI: 10.15406/jpnc.2017.07.00290
- Aricò, M.; Caselli, D. Acute, Severe Hepatitis of Unknown Origin: Should We Really Be Afraid of Another Obscure Enemy of Our Children? Pediatr. Rep. 2022, 14, 217–219. https://doi.org/10.3390/pediatric14020029
- Brodin P, Arditi M. Severe acute hepatitis in children: investigate SARS-CoV-2 superantigens. The Lancet Gastroenterology & Hepatology. 2022 May 14. https://doi.org/10.1016/S2468-1253(22)00166-2
- https://sehatnegeriku.kemkes.go.id/baca/umum/20220513/0539829/kemenkes-temukan-18-orang-dugaan-kasus-hepatitis-akut/
- https://www.haaretz.com/israel-news/israel-examining-12- cases-of-kids-hepatitis-after-who-warning-1.10752779
- https://en.tempo.co/read/1588801/covid-19-endemic-status-waits-for-further-evaluation




Most Commented