September to remember the survival war against sepsis: INA-RESPOND participation in Southeast Asia infectious disease clinical research network and hints update of the 2021’s surviving sepsis campaign
By: Upi Nurhayati, Adhella Menur
Each year on September 13th, people worldwide are encouraged to unite in the fight against sepsis and commemorate it as World Sepsis Day. The day was initiated by a non-profit organization, the Global Sepsis Alliance (GSA), in 2012 to spread sepsis awareness and show solidarity with the millions of people who lost their loved ones, or, as sepsis survivors, suffer from long-term consequences of sepsis.1 The word sepsis is derived from the Greek word for “decay.” Its first documented use was about 2700 years ago in Homer’s poems which described a severely ill man who lay in a tent for 12 days. Hippocratic literature (around 400 BC) described clinical sepsis course as a local lesion, heated by humour afflux, making the whole body feverish, and one can die because of this.2 Decades after decades, scientists and clinicians tried to understand the definition and complex pathophysiology of sepsis. In 2016, the task force team appointed by SCCM (Society of Critical Care Medicine) and ESICM (European Society of Intensive Care Medicine) released the new definition of sepsis known as Sepsis-3: “Sepsis is a life-threatening organ dysfunction caused by dysregulated host response to infection”.3
Sepsis is the final common pathway of all severe infectious diseases, including, most recently, COVID-19. Despite significant advancements in understanding the pathophysiology of clinical sepsis syndrome and intensive care, sepsis remains a leading killer worldwide, accounting for about 20% of all global deaths annually. Furthermore, among adult sepsis survivors, one in three died within a year and one in six experienced significant, long-term morbidity. The sad truth, sepsis is actually preventable through early diagnosis, timely and appropriate treatment, and effective infection prevention and control measures.4

Figure. The dysregulated immune responses in sepsis manifested in poor cellular and tissue barrier function, loss of specialized tissue actions, and a form of cellular hibernation. Individual cells’ behavior changes to unicellular survival mode and becomes functionally incapable, resulting in organ dysfunction. This general hyporeactive survival strategy during cellular stress might preserve cellular energy status and maintain individual cell viability but can be lethal to the host. Therefore, sepsis management aims to achieve a homeostasis state so that every cell can work safely and normally.5
In 2015, INA-RESPOND, together with Thailand and Vietnam research networks, participated in a multinational, multicentre cross-sectional study of community-acquired sepsis in children and adults coordinated by the SEAICRN partner network (Southeast Asia Infectious Disease Clinical Research Network). With a spirit to reduce sepsis mortality, the study was the first that aimed to identify potential causes of sepsis using a predefined set of diagnostic tests covering a wide range of viruses, bacteria, and parasites in Southeast Asia. The study also aimed to determine sepsis management and outcomes. The main study results were published in the Lancet Global Health journal in 2017, entitled “Causes and outcomes of sepsis in southeast Asia: a multinational multicentre cross-sectional study.”6
In Indonesia, the study was conducted at three hospitals (Dr. Cipto Mangunkusumo Hospital, Jakarta, Dr. Sardjito Hospital, Yogyakarta, and Dr. Wahidin Hospital, Makassar). The research assistant enrolled eligible pediatric patients (≥30 days and <18 years) and adult patients (≥18 years) who were admitted with a primary diagnosis of suspected or documented infection, were within 24 h of hospital admission and had at least three sepsis diagnostic criteria documented in the medical record based on the 2012’s surviving sepsis campaign (SSC). The study excluded patients who were suspected of having hospital-acquired infections, had a hospital stay within 30 days before this admission, or were transferred from other hospitals with a total duration of hospitalization of more than 72 h. Participants were observed during enrollment, with the first follow-up visit occurring 14-20 days after enrollment and the last visit occurring 28-35 days after enrollment. At the baseline-enrolment visit, the study team performed data collection, several rapid diagnostic tests (whole blood lactate, dengue NS1 and IgM, influenza, and leptospira IgM/IgG), and biological samples collection (blood in all participants, nasal and throat swabs in patients with respiratory symptoms, stool in patients with diarrhoeal symptoms, and residual cerebrospinal fluid if available). A set of reference diagnostic tests (culture, molecular, and serological tests) was performed for each patient according to clinical presentation. At the first follow-up, the study team performed data collection and convalescent blood samples collection. At the last visit, the study team collected each participant’s survival questionnaires and clinical outcomes.6
The study successfully enrolled 1582 community-acquired sepsis patients (every 750 subjects from Thailand and Vietnam and 82 from Indonesia), consist of 815 adults and 763 pediatrics for analysis. Acute respiratory infection was the most frequent clinical presentation, found in 481 (63%) children and 436 (53%) adults. At least one pathogen was identified in 425 (56%) children and 388 (48%) adults, and one child (<1%) and 17 (2%) adults had a final diagnosis of non-infectious causes. Dengue viruses (n=122 [8%]), Leptospira spp (n=95 [6%]), rickettsial pathogens (n=96 [6%]), Escherichia coli (n=76 [5%]), and influenza viruses (n=65 [4%]) were commonly identified in both age groups. Emerging pathogens identified included hantaviruses (n=28 [2%]), non-typhoidal Salmonella spp (n=21 [1%]), Streptococcus suis (n=18 [1%]), Acinetobacter spp (n=12 [1%]), and Burkholderia pseudomallei (n=5 [<1%]). It should be kept in mind that the decision to perform some diagnostic tests in the study in predefined subsets of patients with relevant symptoms could lead to misdiagnosis of some infectious diseases. Also, the pathogens identified might not be the cause of sepsis in some cases as there is a possibility that even some pathogenic viruses and bacteria could be found in the nasopharyngeal cavity of healthy patients, and background seropositivity for rickettsiosis and leptospirosis is possible in people living in endemic areas.6
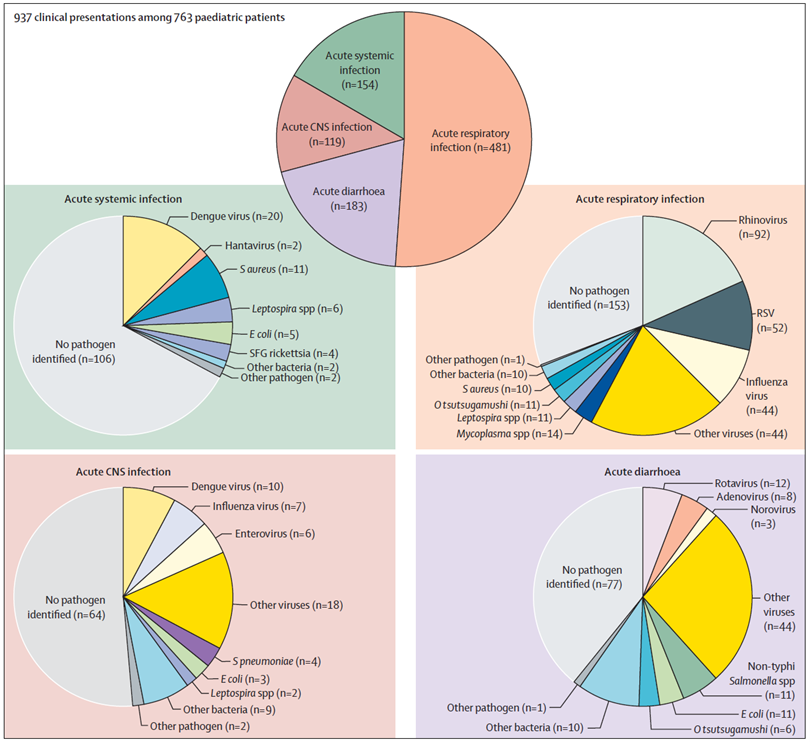
The study results highlighted the diverse causes of sepsis and the need for rapid, inexpensive, and accurate multi-disease diagnostic tests for tropical developing countries. Awareness of the epidemiology of sepsis will help clinicians develop the most appropriate differential to guide empirical treatment. Hence, the first broad-spectrum antibiotics for patients with severe sepsis in southeast Asia need to cover both Gram-negative and Gram-positive bacteria. Doxycycline might need to be included as commonly used empirical antimicrobials such as beta-lactams and carbapenems are not effective against Orientia tsutsugamushi. In areas where melioidosis is endemic, the third-generation cephalosporin might need to be ceftazidime as other third generation cephalosporins are ineffective against B. pseudomallei.6
In the study, adult patients with SOFA scores ≥2 on admission were associated with higher 28-day mortality than adult patients with SOFA scores <2 on admission (99 [22%] of 454 vs. 9 [3%] of 350; adjusted OR 4.1, 95% CI 1.6–10.3; p=0.003). This suggests that patients suspected of infection should be routinely screened for signs of tissue hypoperfusion or organ dysfunction. Patients with sepsis should receive intensive care and organ support immediately to avoid unfavorable outcomes, consistent with the international sepsis task force recommendations.7
In 2018, the Site Investigator from Dr. Cipto Mangunkusumo Hospital, Jakarta, dr. Khie Chen Lie et al. produced a sub-analysis manuscript of 454 sepsis subjects from the parent study which published in Journal of Intensive Care entitled “Utility of SOFA score, management and outcomes of sepsis in Southeast Asia: a multinational multicenter prospective observational study”. The sub-analysis objective was to evaluate management, outcomes, adherence to sepsis bundles based on SSC 2016 recommendations, and mortality prediction of maximum SOFA scores. The main findings were that adherence to SSC bundles even at the late time point of 24 h after admission was generally low: broad-spectrum antibiotics in 76% (344/454), ≥ 1500 mL fluid in 50% of patients with hypotension or lactate ≥ 4 mmol/L (115/231), and adrenergic agents in 71% of patients with hypotension (135/191). Most patients were not admitted to the ICU but were managed on the wards and the 28-day mortality is 22%. Proactive strategies for increasing adherence are necessary and may be uniquely challenging in limited resource settings with an emphasis on efficacy, cost-effectiveness, and feasibility.7
For the most part, SOFA scores can be measured with the available standard of care resources in low-middle-income countries (LMICs) in Southeast Asia with considerate additional costs. However, the SOFA respiration score is based on the PaO2 from an arterial blood gas which is still limited to access. Reframing the SOFA respiratory score for LMICs by using SpO2/FiO2 or simply based on a requirement for supplemental oxygen or respiratory support devices may be helpful. Total SOFA score on admission of those who subsequently died was significantly higher than that of those who survived (6.7 vs. 4.6, p < 0.001). The number of organ failures showed a significant correlation with 28-day mortality, which ranged from 7% in patients without any organ failure to 47% in those with failure of at least four organs (p < 0.001). This association was also valid for individual organ SOFA scores, except for the coagulation score. This may be a spurious finding or due to the wide range of sepsis pathogens with similar presentation but different mechanism and impact in coagulation system, for example thrombocytopenia in bacterial vs. viral causes. To reduce sepsis-related mortality in LMICs, the fundamental elements of sepsis care must undoubtedly be tailored to and evaluated in these settings.7
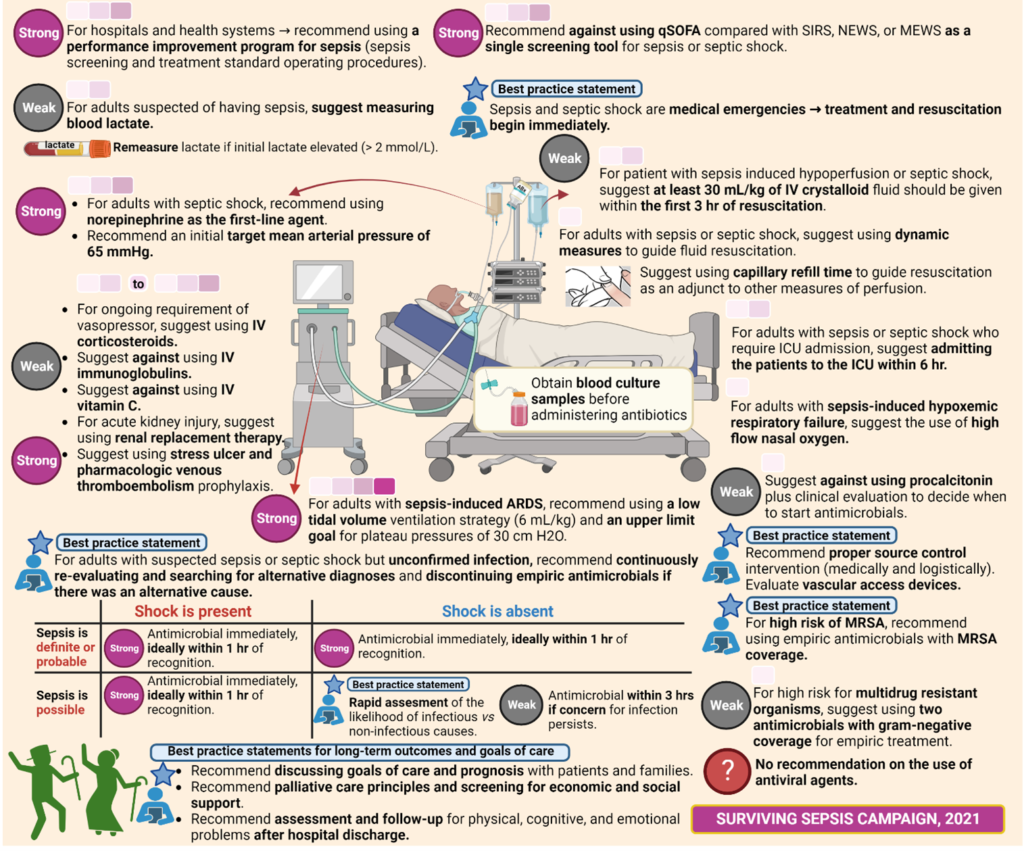
Sepsis is a common and deadly condition, but diagnosis is not always knowable in real-time. To date, no diagnostic tool can rapidly identify or exclude infection, which contributes to the high quantities of broad-spectrum antibiotics used globally (antibiotic resistance threat) and is likely to lead to the under-recognition of non-infectious inflammatory states. Although the sepsis outcomes have been improved in recent years, mortality remains high. No specific anti-sepsis treatments exist; as such, management of patients relies mainly on early recognition allowing correct therapeutic measures to be started rapidly, including administration of appropriate antibiotics, source control measures when necessary, and resuscitation with intravenous fluids and vasoactive drugs when needed. Therefore, identifying and managing sepsis continues to rely on thorough personalized patient assessment and sound clinical judgment. In 2021’s SSC guidelines for the management of sepsis and septic shock, the task force team underscores that the recommendations are intended to reflect best practices and cannot replace the clinician’s decision-making capability when presented with a unique patient’s clinical variables. The recommendations of the SSC 2021 guidelines update are provided in 93 total statements, which address screening and initial resuscitation (n = 10 statements), infection (n = 21), hemodynamics (n = 14), ventilation (n = 12), additional therapies (n = 16), and goals of care and long-term outcomes (n = 20). 8,9,10 Sepsis seems to be stubbornly resisted all efforts to develop and deploy new and improved diagnostic tools and treatments. We believe that we will win the war against sepsis; all we have to do is continuously fight and survive. Stop sepsis, save lives!
More to read:
- https://www.worldsepsisday.org/
- Botero JS, Pérez MC. The history of sepsis from ancient Egypt to the XIX century. InSepsis-an ongoing and significant challenge 2012. Intechopen. doi: 10.5772/51484.
- Singer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016. doi: 10.1001/jama.2016.0287.
- WHO. Global report on the epidemiology and burden of sepsis: current evidence, identifying gaps and future directions. 2020.
- Cohen J, et al. Sepsis: a roadmap for future research. The Lancet infectious diseases. 2015. doi: 10.1016/S1473-3099(15)70112-X.
- Southeast Asia Infectious Disease Clinical Research Network. Causes and outcomes of sepsis in southeast Asia: a multinational multicentre cross-sectional study. The Lancet Global health. 2017. doi: 10.1016/S2214-109X(17)30007-4.
- Lie KC, et al. Utility of SOFA score, management and outcomes of sepsis in Southeast Asia: a multinational multicenter prospective observational study. J of intensive care. 2018. doi: 10.1186/s40560-018-0279-7.
- Prescott HC, Iwashyna TJ. Improving sepsis treatment by embracing diagnostic uncertainty. Annals of the American Thoracic Society. 2019. doi: 10.1513/AnnalsATS.201809-646PS.
- Duncan CF, et al. Diagnostic Challenges in Sepsis. Current Infectious Disease Reports. 2021. doi: 10.1007/s11908-021-00765-y.
- Evans L, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive care medicine. 2021. doi: 10.1007/s00134-021-06506-y.

Most Commented